Thermodynamics(Chemistry)
NCERT Textbook Solution (Laptop/Desktop is best to view this page)
NCERT Solutions for Class 11 Chemistry Chapter 6
Thermodynamics Class 11
Chapter 6 Thermodynamics Exercise Solutions
�Exercise : Solutions of Questions on Page Number : 182���������������������������������������������������������������������������������������������������������������������
Q1 :
Choose the correct answer. A thermodynamic state function is a quantity
(i) used to determine heat changes
(ii) whose value is independent of path
(iii) used to determine pressure volume work
(iv) whose value depends on temperature only.
Answer :
![]() A thermodynamic state function is a quantity whose value
is independent of a path. Functions
like p, V, Tetc. depend only on the state of a system and not on the
path. Hence, alternative (ii) is correct.
A thermodynamic state function is a quantity whose value
is independent of a path. Functions
like p, V, Tetc. depend only on the state of a system and not on the
path. Hence, alternative (ii) is correct.
Q2 :
For the process to occur under adiabatic conditions, the correct condition is:
(i) ΔT = 0
(ii) Δp = 0
(iii) q = 0
(iv) w= 0
Answer :
A system is said to be under adiabatic conditions if there is no exchange of heat between the system and its surroundings. Hence, under adiabatic conditions, q = 0.
Therefore, alternative (iii) is correct.
Q3 :
The enthalpies of all elements in their standard states are:
(i) unity
(ii) zero
(iii) < 0
(iv) different for each element
Answer :
![]() The enthalpy of all elements in their standard state is
zero. Therefore, alternative (ii) is correct.
The enthalpy of all elements in their standard state is
zero. Therefore, alternative (ii) is correct.
Q4 :
ΔUθof combustion of methane is - XkJ mol-1. The value of ΔHθis
(i) = ΔUθ
(ii) > ΔUθ
(iii) < ΔUθ
(iv) = 0
Answer :
SinceΔHθ= ΔUθ+ ΔngRT and ΔUθ= -XkJ mol-1, ΔHθ= (-X) + ΔngRT.
⇒ ΔHθ< ΔUθ
Therefore, alternative (iii) is correct.
Q5 :
The enthalpy of combustion of methane, graphite and dihydrogen at 298 K are, -890.3 kJ mol-1 -393.5 kJ mol-1, and -285.8 kJ mol-1 respectively. Enthalpy of formation of CH4(g)� will be
(i) -74.8 kJ mol-1��������������������� (ii) -52.27 kJ mol-1
(iii) +74.8 kJ mol-1������������������ (iv) +52.26 kJ mol-1.
Answer :
According to the question,
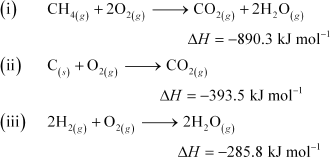
Thus, the desired equation is the one that represents the formation of CH4 (g)i.e.,
![]()
![]() Enthalpy of formation of CH4(g) = –74.8 kJ mol–1 Hence, alternative (i) is correct.
Enthalpy of formation of CH4(g) = –74.8 kJ mol–1 Hence, alternative (i) is correct.
Q6 :
A reaction, A + B → C + D + qis found to have a positive entropy change. The reaction will be
(i) possible at high temperature
(ii) possible only at low temperature
(iii) not possible at any temperature
(iv) possible at any temperature
Answer :
For a reaction to be spontaneous, ΔGshould be negative. ΔG= ΔH- TΔS
According to the question, for the given reaction, ΔS= positive
ΔH= negative (since heat is evolved)
⇒ ΔG= negative
![]() Therefore, the reaction is spontaneous at any
temperature. Hence, alternative (iv) is correct.
Therefore, the reaction is spontaneous at any
temperature. Hence, alternative (iv) is correct.
Q7 :
In a process, 701 J of heat is absorbed by a system and 394 J ofwork is done by the system. What is the change in internal energy for the process?
Answer :
According to the first law of thermodynamics, ΔU= q+ W (i)
Where,
ΔU= change in internal energy for a process
q= heat W= work Given,
q= + 701 J (Since heat is absorbed)
W= -394 J (Since work is done by the system) Substituting the values in expression (i), we get ΔU= 701 J + (-394 J)
ΔU= 307 J
Hence, the change in internal energy for the given process is 307 J.
Q8 :
The reaction of cyanamide, NH2CN(s),with dioxygen was carried out in a bomb calorimeter, and ΔU was found to be -742.7 kJ mol-1at 298 K. Calculate enthalpy change for the reaction at 298 K.
Answer :
Enthalpy change for a reaction (ΔH) is given by the expression, ΔH = ΔU + ΔngRT
Where,
ΔU = change in internal energy Δng = change in number of moles For the given reaction,
Δng = ∠�ng (products) - ∠�ng (reactants)
= (2 - 1.5) moles Δng = 0.5 moles
And,
ΔU = -742.7 kJ mol-1
T = 298 K
R = 8.314 x 10-3 kJ mol-1 K-1
Substituting the values in the expression of ΔH:
ΔH = (-742.7 kJ mol-1) + (0.5 mol) (298 K) (8.314 x 10-3 kJ mol-1 K-1)
= -742.7 + 1.2
ΔH = -741.5 kJ mol-1
Q9 :
Calculate the number of kJ of heat necessary to raise the temperatureof 60.0 g of aluminium from 35�C to 55�C. Molar heat capacity of Al is 24 J mol-1 K-1.
Answer :
From the expression of heat (q),
q = m. c. ΔT Where,
c = molar heat capacity
m = mass of substance
ΔT = change in temperature
Substituting the values in the expression of q:
q = 1066.7 J
q = 1.07 kJ
Q10 :
Calculate the enthalpy change on freezing of 1.0 mol of water at 10.0�C to ice at -10.0�C. ΔfusH = 6.03 kJ mol- 1 at 0�C.
Cp[H2O(l)] = 75.3 J mol-1� K-1
Cp[H2O(s)] = 36.8 J mol-1 K-1
Answer :
Total enthalpy change involved in the transformation is the sum of the following changes:
(a) Energy change involved in the transformation of 1 mol of water at 10�C to 1 mol of water at 0�C.
(b) Energy change involved in the transformation of 1 mol of water at 0� to 1 mol of ice at 0�C.
(c) Energy change involved in the transformation of 1 mol of ice at 0�C to 1 mol of ice at –10�C.
= (75.3 J mol–1 K–1) (0 – 10)K + (–6.03 � 103 J mol–1) + (36.8 J mol–1 K–1) (–10 – 0)K
= –753 J mol–1– 6030 J mol–1– 368 J mol–1
= –7151 J mol–1
= –7.151 kJ mol–1
Hence, the enthalpy change involved in the transformation is –7.151 kJ mol–1.
Q11 :
Enthalpy of combustion of carbon to CO2is -393.5 kJ mol-1. Calculate the heat released upon formation of 35.2 g of CO2from carbon and dioxygen gas.
Answer :
Formation of CO2 from carbon and dioxygen gas can be represented as:
(1 mole = 44 g)
![]() Heat released on formation of 44 g CO2= –393.5 kJ mol–1 Heat released on formation of 35.2 g CO2
Heat released on formation of 44 g CO2= –393.5 kJ mol–1 Heat released on formation of 35.2 g CO2
![]()
= –314.8 kJ mol–1
Q12 :
Enthalpies of formation of CO(g), CO2(g), N2O(g) and N2O4(g) are –110 kJ mol–1, – 393 kJ mol–1, 81 kJ mol–1and
9.7 kJ mol–1respectively. Find the value of ΔrH for the reaction:
![]() N2O4(g)+ 3CO(g)���������������������� N2O(g)+ 3CO2(g)
N2O4(g)+ 3CO(g)���������������������� N2O(g)+ 3CO2(g)
Answer :
ΔrHfor a reaction is defined as the difference between ΔfHvalue of products and ΔfHvalue of reactants.
For the given reaction,
![]() N2O4(g)+ 3CO(g)���������������������� N2O(g)+ 3CO2(g)
N2O4(g)+ 3CO(g)���������������������� N2O(g)+ 3CO2(g)
Substituting the values of ΔfHfor N2O, CO2, N2O4,and CO from the question, we get:
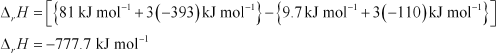 |
Q13 :
Given
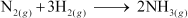 ; ΔrHθ= –92.4 kJ mol–1 What is the standard
enthalpy of formation of NH3gas?
; ΔrHθ= –92.4 kJ mol–1 What is the standard
enthalpy of formation of NH3gas?
Answer :
Standard enthalpy of formation of a compound is the change in enthalpy that takes place during the formation of 1 mole of a substance in its standard form from its constituent elements in their standard state.
Re-writing the given equation for 1 mole of NH3(g),
![]() Standard enthalpy
of formation of NH3(g)
Standard enthalpy
of formation of NH3(g)
= � ΔrHθ
= � (–92.4 kJ mol–1)
= –46.2 kJ mol–1
Q14 :
![]() Calculate the standard
enthalpy of formation of CH3OH(l)� from the following
data:
Calculate the standard
enthalpy of formation of CH3OH(l)� from the following
data:
![]() CH3OH(l) +������ O2(g)������������������������ CO2(g)
+
2H2O(l) ; ΔrHθ = –726 kJ mol–1
CH3OH(l) +������ O2(g)������������������������ CO2(g)
+
2H2O(l) ; ΔrHθ = –726 kJ mol–1
![]() C(g) +
O2(g)������������������������ CO2(g) ;
ΔcHθ = –393
kJ mol–1 H2(g) +
C(g) +
O2(g)������������������������ CO2(g) ;
ΔcHθ = –393
kJ mol–1 H2(g) +
Answer :
![]() The reaction that takes place during the formation of CH3OH(l)can be written
as:
The reaction that takes place during the formation of CH3OH(l)can be written
as:
![]() C(s)+
2H2O(g)+������ O2(g)����������������������� CH3OH(l)� (1)
C(s)+
2H2O(g)+������ O2(g)����������������������� CH3OH(l)� (1)
The reaction (1) can be obtained from the given reactions by following the algebraic calculations as: Equation (ii) + 2 � equation (iii) – equation (i)
ΔfHθ[CH3OH(l)] = ΔcHθ + 2ΔfHθ[H2O(l)] – ΔrHθ
= (–393 kJ mol–1) + 2(–286 kJ mol–1) – (–726 kJ mol–1)
![]()
![]() = (–393 – 572 + 726) kJ mol–1 ΔfHθ[CH3OH(l)]
= –239 kJ mol–1
= (–393 – 572 + 726) kJ mol–1 ΔfHθ[CH3OH(l)]
= –239 kJ mol–1
Q15 :
Calculate the enthalpy change for the process CCl4(g) → C(g)� + 4Cl(g)
and calculate bond enthalpy of C-Cl in CCl4(g). ΔvapHθ (CCl4) = 30.5 kJ mol-1.
ΔfHθ (CCl4) = -135.5 kJ mol-1.
ΔaHθ (C) = 715.0 kJ mol-1, where ΔaHθ is enthalpy of atomisation ΔaHθ (Cl2) = 242 kJ mol-1
Answer :
![]() The chemical equations
implying to the given values
of enthalpies are:
The chemical equations
implying to the given values
of enthalpies are:
![]()
![]() ΔvapHθ = 30.5 kJ mol–1 ΔaHθ = 715.0 kJ mol–1
ΔvapHθ = 30.5 kJ mol–1 ΔaHθ = 715.0 kJ mol–1
![]() ΔaHθ = 242 kJ
mol–1
ΔaHθ = 242 kJ
mol–1
ΔfH = –135.5 kJ mol–1
![]() Enthalpy change for the given process������������������������������������������������������������� can
be calculated using the following algebraic calculations as:
Enthalpy change for the given process������������������������������������������������������������� can
be calculated using the following algebraic calculations as:
Equation (ii) + 2 � Equation (iii) – Equation (i) – Equation (iv) ΔH = ΔaHθ(C) + 2ΔaHθ (Cl2) – ΔvapHθ – ΔfH
= (715.0 kJ mol–1) + 2(242 kJ mol–1) – (30.5 kJ mol–1) – (–135.5 kJ mol–1)
![]() ΔH = 1304 kJ
mol–1
ΔH = 1304 kJ
mol–1
Bond enthalpy of C–Cl bond in CCl4 (g)
= 326 kJ mol–1
Q16 :
For an isolated system, ΔU = 0, what will be ΔS?
Answer :
ΔS will be positive i.e., greater than zero
Since ΔU= 0, ΔSwill be positive and the reaction will be spontaneous.
Q17 :
For the reaction at 298 K, 2A + B → C
ΔH = 400 kJ mol-1and ΔS = 0.2 kJ K-1mol-1
At what temperature will the reaction become spontaneous considering ΔH and ΔS to be constant over the temperature range?
Answer :
From the expression, ΔG= ΔH– TΔS
Assuming the reaction at equilibrium, ΔTfor the reaction would be:
![]() (ΔG = 0 at equilibrium)
(ΔG = 0 at equilibrium)
T= 2000 K
For the reaction to be spontaneous, ΔGmust be negative. Hence, for the given reaction to be spontaneous, Tshould be greater than 2000 K.
Q18 :
For the reaction,
2Cl(g) → Cl2(g),what are the signs of ΔH and ΔS ?
Answer :
ΔHand ΔS are negative
The given reaction represents the formation of chlorine molecule from chlorine atoms. Here, bond formation is taking place. Therefore, energy is being released. Hence, ΔHis negative.
Also, two moles of atoms have more randomness than one mole of a molecule. Since spontaneity is decreased, ΔSis negative for the given reaction.
Q19 :
For the reaction 2A(g) + B(g) → 2D(g)
ΔUθ = -10.5 kJ and ΔSθ= -44.1 JK-1.
Calculate ΔGθ for the reaction, and predict whether the reaction may occur spontaneously.
Answer :
For the given reaction, 2 A(g) + B(g) → 2D(g)
Δng = 2 - (3)
= -1 mole
Substituting the value of ΔUθ in the expression of ΔH:
ΔHθ = ΔUθ + ΔngRT
= (-10.5 kJ) - (-1) (8.314 x 10-3 kJ K-1 mol-1) (298 K)
= -10.5 kJ - 2.48 kJ ΔHθ = -12.98 kJ
Substituting the values of ΔHθ and ΔSθ in the expression of ΔGθ: ΔGθ = ΔHθ - TΔSθ
= -12.98 kJ - (298 K) (-44.1 J K-1)
= -12.98 kJ + 13.14 kJ ΔGθ = + 0.16 kJ
Since ΔGθ for the reaction is positive, the reaction will not occur spontaneously.
Q20 :
The equilibrium constant for a reaction is 10. What will be the valueof ΔGθ? R = 8.314 JK-1mol-1, T= 300 K.
Answer :
From the expression, ΔGθ= -2.303 RTlogKeq
ΔGθfor the reaction,
= (2.303) (8.314 JK-1mol-1) (300 K) log10
= -5744.14 Jmol-1
= -5.744 kJ mol-1
Q21 :
![]()
![]() Comment on the thermodynamic stability of NO(g),given
Comment on the thermodynamic stability of NO(g),given
![]() N2(g)
+������ O2(g) â� � NO(g); ΔrHθ= 90 kJ mol–1
N2(g)
+������ O2(g) â� � NO(g); ΔrHθ= 90 kJ mol–1
NO(g)+����� O2(g) â� � NO2(g)� : ΔrHθ= –74 kJ mol–1
Answer :
The positive value of ΔrH indicates that heat is absorbed during the formation of NO(g). This means that NO(g) has higher energy than the reactants (N2 and O2). Hence, NO(g) is unstable.
The negative value of ΔrH indicates that heat is evolved during the formation of NO2(g) from NO(g) and O2(g). The product, NO2(g) is stabilized with minimum energy.
Hence, unstable NO(g)� changes to stable NO2(g).
Q22 :
Calculate the entropy change in surroundings when 1.00 mol of H2O(l)is formed under standard conditions. ΔfHθ= -286 kJ mol-1.
Answer :
It is given that 286 kJ mol–1of heat is evolved on the formation of 1 mol of H2O(l). Thus, an equal amount of heat will be absorbed by the surroundings.
qsurr= +286 kJ mol–1
![]() Entropy change (ΔSsurr) for the surroundings =
Entropy change (ΔSsurr) for the surroundings =
![]() ΔSsurr=
959.73 J mol–1K–1
ΔSsurr=
959.73 J mol–1K–1