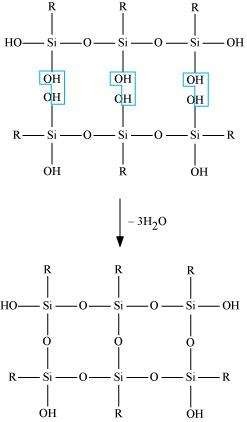
The p-Block Elements(11)
NCERT Textbook Solution (Laptop/Desktop is best to view this page)
NCERT Solutions for Class 11 Chemistry Chapter 11
The p-Block Elements Class 11
Chapter 11 The p-Block Elements Exercise Solutions
�Exercise : Solutions of Questions on Page Number : 323���������������������������������������������������������������������������������������������������������������������
Q1 :
Discuss the pattern of variation in the oxidation states of
(i) B to Tl and (ii) C to Pb.
Answer :
(i) B to Tl
The electric configuration of group 13 elements is ns2 np1. Therefore, the most common oxidation state exhibited by them should be +3. However, it is only boron and aluminium which practically show the +3 oxidation state. The remaining elements, i.e., Ga, In, Tl, show both the +1 and +3 oxidation states. On moving down the group, the +1 state becomes more stable. For example, Tl (+1) is more stable than Tl (+3). This is because of the inert pair effect. The two electrons present in the s-shell are strongly attracted by the nucleus and do not participate in bonding. This inert pair effect becomes more and more prominent on moving down the group. Hence, Ga (+1) is unstable, In (+1) is fairly stable, and Tl (+1) is very stable.
|
Group 13 element |
Oxidation state |
|
B |
+3 |
|
Al |
+3 |
|
Ga, In, Tl |
+1, +3 |
The stability of the +3 oxidation state decreases on moving down the group.
(ii) C to Pb
The electronic configuration of group 14 elements is ns2 np2. Therefore, the most common oxidation state exhibited by them should be +4. However, the +2 oxidation state becomes more and more common on moving down the group. C and Si mostly show the +4 state. On moving down the group, the higher oxidation state becomes less stable. This is because of the inert pair effect. Thus, although Ge, Sn, and Pb show both the +2 and + 4 states, the stability of the lower oxidation state increases and that of the higher oxidation state decreases on moving down the group.
|
Group 14 element |
Oxidation state |
|
C |
+4 |
|
Si |
+4 |
|
Ge, Sn, Pb |
+2, +4 |
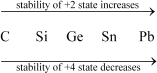
Q2 :
How can you explain higher stability of BCl3 as compared to TlCl3?
Answer :
Boron and thallium belong to group 13 of the periodic table. In this group, the +1 oxidation state becomes more stable on moving down the group. BCl3is more stable than TlCl3because the +3 oxidation state of B is more stable than the
+3 oxidation state of Tl. In Tl, the +3 state is highly oxidising and it reverts back to the more stable +1 state.
Q3 :
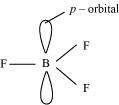
Why does boron trifluoride behave as a Lewis acid?
Answer :
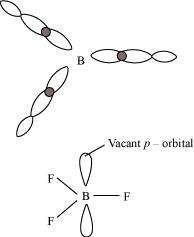 |
The electric configuration of boron is ns2 np1. It has three electrons in its valence shell. Thus, it can form only three covalent bonds. This means that there are only six electrons around boron and its octet remains incomplete. When one atom of boron combines with three fluorine atoms, its octet remains incomplete. Hence, boron trifluoride remains electron-deficient and acts as a Lewis acid.
![]()
Q4 :
Consider the compounds, BCl3 and CCl4. How will they behave with water? Justify.
Answer :
Being a Lewis acid, BCl3 readily undergoes hydrolysis. Boric acid is formed as a result.
CCl4completely resists hydrolysis. Carbon does not have any vacant orbital. Hence, it cannot accept electrons from water to form an intermediate. When CCl4and water are mixed, they form separate layers.
Q5 :
Is boric acid a protic acid? Explain.
Answer :
Boric acid is not a protic acid. It is a weak monobasic acid, behaving as a Lewis acid.
It behaves as an acid by accepting a pair of electrons from –OH ion.
![]()
Q6 :
Explain what happens when boric acid is heated.
Answer :
On heating orthoboric acid (H3BO3) at 370 K or above, it changes to metaboric acid (HBO2). On further heating, this yields boric oxide B2O3.
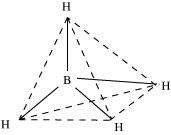
Q7 :
4
Describe the shapes of BF3and BH -.
Assign the hybridisation of boron in these species.
Answer :
(i) BF3
As a result of its small size and high electronegativity, boron tends to form monomeric covalent halides. These halides have a planar triangular geometry. This triangular shape is formed by the overlap of three sp2hybridised orbitals of boron with the sporbitals of three halogen atoms. Boron is sp2hybridised in BF3.
(ii)
4
BH -
 |
|
![]()
Q8 :
Write reactions to justify amphoteric nature of aluminium.
Answer :
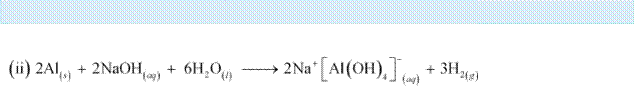 |
A substance is called amphoteric if it displays characteristics of both acids and bases. Aluminium dissolves in both acids and bases, showing amphoteric behaviour.
Q9 :
What are electron deficient compounds? Are BCl3and SiCl4electron deficient species? Explain.
Answer :
In an electron-deficient compound, the octet of electrons is not complete, i.e., the central metal atom has an incomplete octet. Therefore, it needs electrons to complete its octet.
(i) BCl3
BCl3is an appropriate example of an electron-deficient compound. B has 3 valence electrons. After forming three covalent bonds with chlorine, the number of electrons around it increases to 6. However, it is still short of two electrons to complete its octet.
(ii) SiCl4
The electronic configuration of silicon is ns2 np2. This indicates that it has four valence electrons. After it forms four covalent bonds with four chlorine atoms, its electron count increases to eight. Thus, SiCl4is not an electron-deficient compound.
Q10 :
![]() Write the resonance structures of
Write the resonance structures of ![]() �and
�and ![]() .
Answer :
.
Answer :
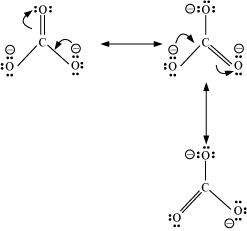 |
(a)
(b)�![]()
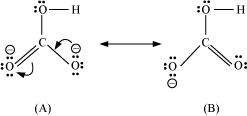
There are only two resonating structures for the bicarbonate ion.
Q11 :
![]() What is the state
of hybridisation of carbon in (a)�������������� (b) diamond
(c) graphite?
What is the state
of hybridisation of carbon in (a)�������������� (b) diamond
(c) graphite?
Answer :
The state of hybridisation of carbon in:
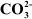 (a)
(a)
![]() C in�������������� is sp2hybridised and is
bonded to three oxygen atoms.
C in�������������� is sp2hybridised and is
bonded to three oxygen atoms.
(b) Diamond
Each carbon in diamond is sp3 hybridised and is bound to four other carbon atoms.
(c) Graphite
Each carbon atom in graphite is sp2hybridised and is bound to three other carbon atoms.
Q12 :
Explain the difference in properties of diamond and graphite on the basis of their structures.
Answer :
|
Diamond |
Graphite |
|
|
It has a crystalline lattice. |
It has a layered structure. |
|
|
In diamond, each carbon atom is sp3� �hybridised and is bonded to four other carbon atoms through a �ƒ bond. |
In graphite, each carbon atom is sp2� �hybridised and is bonded to three other carbon atoms through a �ƒ bond. The fourth electron forms a π bond. |
|
|
It is made up of tetrahedral units. |
It has a planar geometry. |
|
|
The C–C bond length in diamond is 154 pm. |
The C–C bond length in graphite is 141.5 pm. |
|
|
It has a rigid covalent bond network which is difficult to break. |
It is quite soft and its layers can be separated easily. |
|
|
It acts as an electrical insulator. |
It is a good conductor of electricity. |
|
|
|
|
|
Q13 :
Rationalise the given statements and give chemical reactions:
�� Lead(II) chloride reacts with Cl2to give PbCl4.
�� Lead(IV) chloride is highly unstable towards heat. �� Lead is known not to form an iodide, PbI4.
Answer :
(a) Lead belongs to group 14 of the periodic table. The two oxidation states displayed by this group is +2 and +4. On moving down the group, the +2 oxidation state becomes more stable and the +4 oxidation state becomes less stable.
This is because of the inert pair effect. Hence, PbCl4is much less stable than PbCl2. However, the formation of PbCl4takes place when chlorine gas is bubbled through a saturated solution of PlCl2.
(b)
On moving down group IV, the higher oxidation state becomes unstable because of the inert pair effect. Pb(IV) is highly unstable and when heated, it reduces to Pb(II).
(c) Lead is known not to form PbI4. Pb (+4) is oxidising in nature and I-is reducing in nature. A combination of Pb(IV) and iodide ion is not stable. Iodide ion is strongly reducing in nature. Pb(IV) oxidises I–to I2and itself gets reduced to Pb(II).
Q14 :
![]() Suggest reasons why the B–F bond lengths
in BF3(130 pm) and������������ (143 pm) differ.
Suggest reasons why the B–F bond lengths
in BF3(130 pm) and������������ (143 pm) differ.
Answer :
 |
This double-bond character causes the bond length to shorten in BF3(130 pm). However, when BF3coordinates with
 |
![]()
Q15 :
If B-Cl bond has a dipole moment, explain why BCl3molecule has zero dipole moment.
Answer :
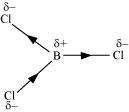 |
As a result of the difference in the electronegativities of B and Cl, the B-Cl bond is polar in nature. However, the BCl3molecule is non-polar. This is because BCl3is trigonal planar in shape. It is a symmetrical molecule. Hence, the respective dipole-moments of the B-Cl bond cancel each other, thereby causing a zero-dipole moment.
![]()
Q16 :
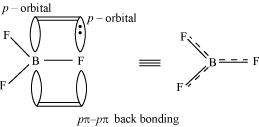
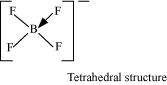
Aluminium trifluoride is insoluble in anhydrous HF but dissolves on addition of NaF. Aluminium trifluoride precipitates out of the resulting solution when gaseous BF3is bubbled through. Give reasons.
Answer :
Hydrogen fluoride (HF) is a covalent compound and has a very strong intermolecular hydrogen-bonding. Thus, it does not provide ions and aluminium fluoride (AlF) does not dissolve in it. Sodium fluoride (NaF) is an ionic compound and when it is added to the mixture, AlF dissolves. This is because of the availability of free F–. The reaction involved in the process is:
When boron trifluoride (BF3) is added to the solution, aluminium fluoride precipitates out of the solution. This happens because the tendency of boron to form complexes is much more than that of aluminium. Therefore, when BF3is added to the solution, B replaces Al from the complexes according to the following reaction:
Q17 :
Suggest a reason as to why CO is poisonous.
Answer :
Carbon monoxide is highly-poisonous because of its ability to form a complex with haemoglobin. The CO-Hb complex is more stable than the O2-Hb complex. The former prevents Hb from binding with oxygen. Thus, a person dies because of suffocation on not receiving oxygen. It is found that the
CO-Hb complex is about 300 times more stable than the O2-Hb complex.
Q18 :
How is excessive content of CO2 responsible for global warming?
Answer :
Carbon dioxide is a very essential gas for our survival. However, an increased content of CO2in the atmosphere poses a serious threat. An increment in the combustion of fossil fuels, decomposition of limestone, and a decrease in the number of trees has led to greater levels of carbon dioxide. Carbon dioxide has the property of trapping the heat provided by sunrays. Higher the level of carbon dioxide, higher is the amount of heat trapped. This results in an increase in the atmospheric temperature, thereby causing global warming.
Q19 :
Explain structures of diborane and boric acid.
Answer :
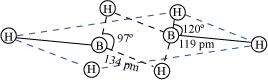
(a) Diborane
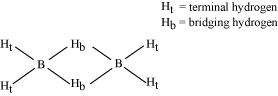 |
B2H6is an electron-deficient compound. B2H6has only 12 electrons - 6 e-from 6 H atoms and 3 e-each from 2 B atoms. Thus, after combining with 3 H atoms, none of the boron atoms has any electrons left. X-ray diffraction studies have shown the structure of diborane as:
2 boron and 4 terminal hydrogen atoms (Ht) lie in one plane, while the other two bridging hydrogen atoms (Hb) lie in a plane perpendicular to the plane of boron atoms. Again, of the two bridging hydrogen atoms, one H atom lies above the plane and the other lies below the plane. The terminal bonds are regular two-centre two-electron (2c - 2e-) bonds, while the two bridging (B-H-B) bonds are three-centre two-electron (3c - 2e-) bonds.
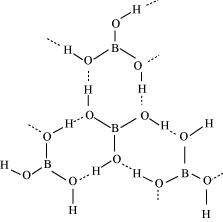
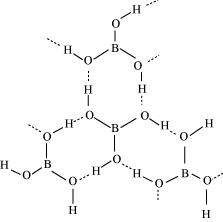
(b) Boric acid
Boric acid has a layered structure. Each planar BO3unit is linked to one another through H atoms. The H atoms form a covalent bond with a BO3 unit, while a hydrogen bond is formed with another BO3unit. In the given figure, the dotted lines represent hydrogen bonds.
![]()
Q20 :
What happens when
(a) Borax is heated strongly,
(b) Boric acid is added to water,
(c) Aluminium is treated with dilute NaOH,
(d) BF3is reacted with ammonia?
Answer :
(a)
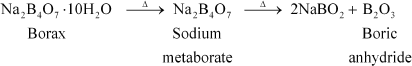 |
When heated, borax undergoes various transitions. It first loses water molecules and swells. Then, it turns into a transparent liquid, solidifying to form a glass-like material called borax bead.
(b) When boric acid is added to water, it accepts electrons from –OH ion.
(c) Al reacts with dilute NaOH to form sodium tetrahydroxoaluminate(III). Hydrogen gas is liberated in the process.
(d) BF3(a Lewis acid) reacts with NH3(a Lewis base) to form an adduct. This results in a complete octet around B in BF3.
Q21 :
Explain the following reactions
(a) Silicon is heated with methyl chloride at high temperature in the presence of copper;
(b) Silicon dioxide is treated with hydrogen fluoride;
(c) CO is heated with ZnO;
(d) Hydrated alumina is treated with aqueous NaOH solution.
Answer :
(a)
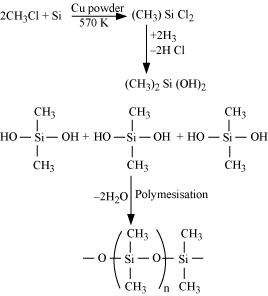 |
When silicon reacts with methyl chloride in the presence of copper (catalyst) and at a temperature of about 537 K, a class of organosilicon polymers called methyl-substituted chlorosilanes (MeSiCl3, Me2SiCl2, Me3SiCl, and Me4Si) are formed.
(b) When silicon dioxide (SiO2) is heated with hydrogen fluoride (HF), it forms silicon tetrafluoride (SiF4). Usually, the Si–O bond is a strong bond and it resists any attack by halogens and most acids, even at a high temperature. However, it is attacked by HF.
The SiF4formed in this reaction can further react with HF to form hydrofluorosilicic acid.
(c) When CO reacts with ZnO, it reduces ZnO to Zn. CO acts as a reducing agent.
(d) When hydrated alumina is added to sodium hydroxide, the former dissolves in the latter because of the formation of sodium meta-aluminate.
Q22 :
Give reasons:
(i) Conc. HNO3can be transported in aluminium container.
(ii) A mixture of dilute NaOH and aluminium pieces is used to open drain.
(iii) Graphite is used as lubricant.
(iv) Diamond is used as an abrasive.
(v) Aluminium alloys are used to make aircraft body.
(vi) Aluminium utensils should not be kept in water overnight.
(vii) Aluminium wire is used to make transmission cables.
Answer :
(i) Concentrated HNO3can be stored and transported in aluminium containers as it reacts with aluminium to form a thin protective oxide layer on the aluminium surface. This oxide layer renders aluminium passive.
(ii)
Sodium hydroxide and aluminium react to form sodium tetrahydroxoaluminate(III) and hydrogen gas. The pressure of the produced hydrogen gas is used to open blocked drains.
(iii) Graphite has a layered structure and different layers of graphite are bonded to each other by weak van der Waals' forces. These layers can slide over each other. Graphite is soft and slippery. Therefore, graphite can be used as a lubricant.
(iv) In diamond, carbon is sp3hybridised. Each carbon atom is bonded to four other carbon atoms with the help of strong covalent bonds. These covalent bonds are present throughout the surface, giving it a very rigid 3-D structure. It is very difficult to break this extended covalent bonding and for this reason, diamond is the hardest substance known. Thus, it is used as an abrasive and for cutting tools.
(v) Aluminium has a high tensile strength and is very light in weight. It can also be alloyed with various metals such as Cu, Mn, Mg, Si, and Zn. It is very malleable and ductile. Therefore, it is used in making aircraft bodies.
(vi) The oxygen present in water reacts with aluminium to form a thin layer of aluminium oxide. This layer prevents aluminium from further reaction. However, when water is kept in an aluminium vessel for long periods of time, some amount of aluminium oxide may dissolve in water. As aluminium ions are harmful, water should not be stored in aluminium vessels overnight.
(vii)
Silver, copper, and aluminium are among the best conductors of electricity. Silver is an expensive metal and silver wires are very expensive. Copper is quite expensive and is also very heavy. Aluminium is a very ductile metal. Thus, aluminium is used in making wires for electrical conduction.
Q23 :
Explain why is there a phenomenal decrease in ionisation enthalpy from carbon to silicon?
Answer :
Ionisation enthalpy of carbon (the first element of group 14) is very high (1086 kJ/mol). This is expected owing to its small size. However, on moving down the group to silicon, there is a sharp decrease in the enthalpy (786 kJ). This is because of an appreciable increase in the atomic sizes of elements on moving down the group.
Q24 :
How would you explain the lower atomic radius of Ga as compared to Al?
Answer :
|
Atomic radius (in pm) |
|
|
Aluminium |
143 |
|
Gallium |
135 |
Although Ga has one shell more than Al, its size is lesser than Al. This is because of the poor shielding effect of the 3d-electrons. The shielding effect of d-electrons is very poor and the effective nuclear charge experienced by the valence electrons in gallium is much more than it is in the case of Al.
Q25 :
What are allotropes? Sketch the structure of two allotropes of carbon namely diamond and graphite. What is the impact of structure on physical properties of two allotropes?
Answer :
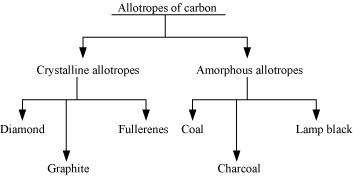 |
Allotropy is the existence of an element in more than one form, having the same chemical properties but different physical properties. The various forms of an element are called allotropes.
Diamond:
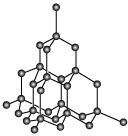
The rigid 3-D structure of diamond makes it a very hard substance. In fact, diamond is one of the hardest naturally- occurring substances. It is used as an abrasive and for cutting tools.
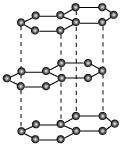 |
Graphite:
It has sp2hybridised carbon, arranged in the form of layers. These layers are held together by weak van der Walls' forces. These layers can slide over each other, making graphite soft and slippery. Therefore, it is used as a lubricant.
![]()
Q26 :
(a) Classify following oxides as neutral, acidic, basic or amphoteric:
CO, B2O3, SiO2, CO2, Al2O3, PbO2, Tl2O3
(b) Write suitable chemical equations to show their nature.
Answer :
(1) CO = Neutral
(2) B2O3= Acidic
Being acidic, it reacts with bases to form salts. It reacts with NaOH to form sodium metaborate.
(3) SiO2 = Acidic
Being acidic, it reacts with bases to form salts. It reacts with NaOH to form sodium silicate.
(4) CO2= Acidic
Being acidic, it reacts with bases to form salts. It reacts with NaOH to form sodium carbonate.
(5) Al2O3= Amphoteric
Amphoteric substances react with both acids and bases. Al2O3reacts with both NaOH and H2SO4.
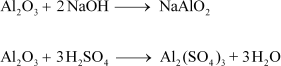 |
(6) PbO2= Amphoteric
Amphoteric substances react with both acids and bases. PbO2reacts with both NaOH and H2SO4.
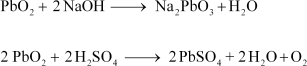 |
(7) Tl2O3= Basic
Being basic, it reacts with acids to form salts. It reacts with HCl to form thallium chloride.
Q27 :
In some of the reactions thallium resembles aluminium, whereas in others it resembles with group I metals. Support this statement by giving some evidences.
Answer :
Thallium belongs to group 13 of the periodic table. The most common oxidation state for this group is +3. However, heavier members of this group also display the +1 oxidation state. This happens because of the inert pair effect.
Aluminium displays the +3 oxidation state and alkali metals display the +1 oxidation state. Thallium displays both the oxidation states. Therefore, it resembles both aluminium and alkali metals.
Thallium, like aluminium, forms compounds such as TlCl3and Tl2O3. It resembles alkali metals in compounds Tl2O and TlCl.
Q28 :
When metal X is treated with sodium hydroxide, a white precipitate (A) is obtained, which is soluble in excess of NaOH to give soluble complex (B). Compound (A) is soluble in dilute HCl to form compound (C). The compound (A) when heated strongly gives (D), which is used to extract metal. Identify (X), (A), (B), (C) and (D). Write suitable equations to support their identities.
Answer :
The given metal X gives a white precipitate with sodium hydroxide and the precipitate dissolves in excess of sodium hydroxide. Hence, X must be aluminium.
The white precipitate (compound A) obtained is aluminium hydroxide. The compound B formed when an excess of the base is added is sodium tetrahydroxoaluminate(III).
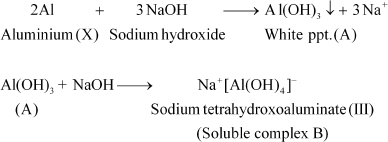 |
Now, when dilute hydrochloric acid is added to aluminium hydroxide, aluminium chloride (compound C) is obtained.
Also, when compound A is heated strongly, it gives compound D. This compound is used to extract metal X. Aluminium metal is extracted from alumina. Hence, compound D must be alumina.
![]()
Q29 :
What do you understand by (a) inert pair effect (b) allotropy and(c) catenation?
Answer :
(a) Inert pair effect
As one moves down the group, the tendency of s-block electrons to participate in chemical bonding decreases. This effect is known as inert pair effect. In case of group 13 elements, the electronic configuration is ns2 np1 and their group valency is +3. However, on moving down the group, the +1 oxidation state becomes more stable. This happens because of the poor shielding of the ns2electrons by the d- and f- electrons. As a result of the poor shielding,
the ns2electrons are held tightly by the nucleus and so, they cannot participate in chemical bonding.
(b) Allotropy
Allotropy is the existence of an element in more than one form, having the same chemical properties but different physical properties. The various forms of an element are called allotropes. For example, carbon exists in three allotropic forms: diamond, graphite, and fullerenes.
(c) Catenation
The atoms of some elements (such as carbon) can link with one another through strong covalent bonds to form long chains or branches. This property is known as catenation. It is most common in carbon and quite significant in Si and S.
Q30 :
A certain salt X, gives the following results.
(i) Its aqueous solution is alkaline to litmus.
(ii) It swells up to a glassy material Yon strong heating.
(iii) When conc. H2SO4is added to a hot solution of X, white crystal of an acid Zseparates out. Write equations for all the above reactions and identify X, Y and Z.
Answer :
The given salt is alkaline to litmus. Therefore, Xis a salt of a strong base and a weak acid. Also, when Xis strongly heated, it swells to form substance Y. Therefore, Xmust be borax.
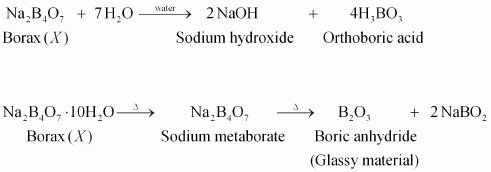 |
When borax is heated, it loses water and swells to form sodium metaborate. When heating is continued, it solidifies to form a glassy material Y. Hence, Ymust be a mixture of sodium metaborate and boric anhydride.
When concentrated acid is added to borax, white crystals of orthoboric acid (Z) are formed.
![]()
![]()
Q31 :
Write balanced equations for:
(i) BF3 + LiH â� �
(ii) B2H6 + H2O â� �
(iii) NaH + B2H6 â� �
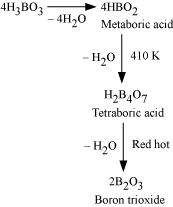
(iv)
H3BO3 ![]()
(v) Al + NaOH â� �
(vi) B2H6 + NH3 â� �
Answer :
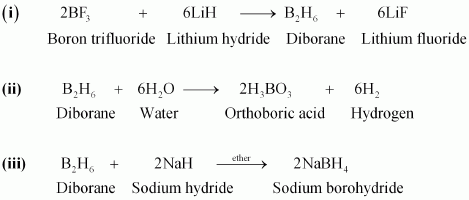
(iv)
 |
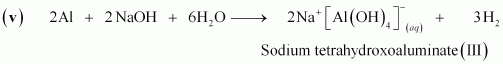
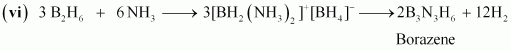 |
![]()
Q32 :
Give one method for industrial preparation and one for laboratory preparation of CO and CO2 each.
Answer :
Caron dioxide
In the laboratory, CO2can be prepared by the action of dilute hydrochloric acid on calcium carbonate. The reaction involved is as follows:
CO2is commercially prepared by heating limestone. The reaction involved is as follows:
![]()
Caron monoxide
In the laboratory, CO is prepared by the dehydration of formic acid with conc. H2SO4, at 373 K. The reaction involved is as follows:
CO is commercially prepared by passing steam over hot coke. The reaction involved is as follows:
![]()
![]()
Q33 :
An aqueous solution of borax is
(a) neutral (b) amphoteric
(c) basic (d) acidic
Answer :
(c) Borax is a salt of a strong base (NaOH) and a weak acid (H3BO3). It is, therefore, basic in nature.
Q34 :
Boric acid is polymeric due to
(a) its acidic nature (b) the presence of hydrogen bonds
(c) its monobasic nature (d) its geometry
Answer :
(b) Boric acid is polymeric because of the presence of hydrogen bonds. In the given figure, the dotted lines represent hydrogen bonds.
![]()
Q35 :
The type of hybridisation of boron in diborane is
(a) sp (b) sp2(c) sp3 (d) dsp2
Answer :
(c) Boron in diborane is sp3hybridised.
Q36 :
Thermodynamically the most stable form of carbon is
(a) diamond (b) graphite
(c) fullerenes (d) coal
Answer :
(b) Graphite is thermodynamically the most stable form of carbon.
Q37 :
Elements of group 14
(a) exhibit oxidation state of +4 only
(b) exhibit oxidation state of +2 and +4
(c) form M2-and M4+ion
(d) form M2+and M4+ions
Answer :
(b)The elements of group 14 have 4 valence electrons. Therefore, the oxidation state of the group is +4. However, as a result of the inert pair effect, the lower oxidation state becomes more and more stable and the higher oxidation state becomes less stable. Therefore, this group exhibits +4 and +2 oxidation states.
|
|
Group 14 element |
Oxidation state |
|
|
C |
+4 |
||
|
Si |
+4 |
||
|
Ge, Sn, Pb |
+2, +4 |
||
|
|
|
||
Q38 :
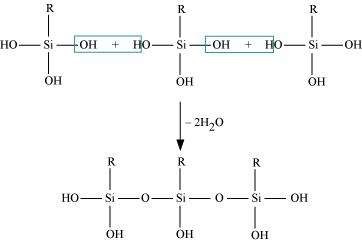
If the starting material for the manufacture of silicones is RSiCl3, write the structure of the product formed.
Answer :
(i)
(ii)