Chemistry in Everyday Life
NCERT Textbook Solution (Laptop/Desktop is best to view this page)
NCERT Solutions for Class 12 Chemistry Part 1 Chapter 16
Chemistry in Everyday Life Class 12
Chapter 16 Chemistry in Everyday Life Exercise Solutions
�In text : Solutions of Questions on Page Number : 448������������������������������������������������������������������������������������������������������������������������
Q1 :
Sleeping pills are recommended by doctors to the patients suffering from sleeplessness but it is not advisable to take its doses without consultation with the doctor, Why?
Answer :
Most drugs when taken in doses higher than recommended may cause harmful effects and sometimes, may even lead to death. Hence, a doctor should always be consulted before taking any medicine.
Q2 :
With reference to which classification has the statement, 'ranitidine is an antacid" been given?
Answer :
The given statement refers to the classification of pharmacological effects of the drug. This is because any drug that is used to counteract the effects of excess acid in the stomach is called an antacid.
Q3 :
Why do we require artificial sweetening agents?
Answer :
A large number of people are suffering from diseases such as diabetes and obesity. These people cannot take normal sugar i.e., sucrose as it is harmful for them. Therefore, artificial sweetening agents that do not add to the calorie intake of a person are required. Saccharin, aspartame, and alitame are a few examples of artificial sweeteners.
Q4 :
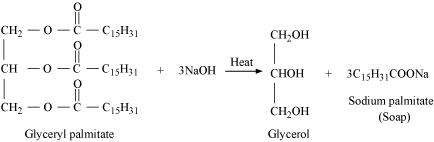
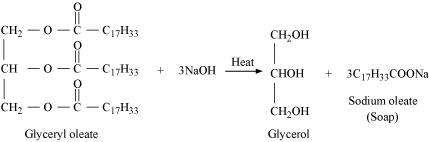
![]() Write the chemical
equation for preparing sodium soap from glyceryl oleate
and glyceryl palmitate. Structural formulae of these compounds
are given below.
Write the chemical
equation for preparing sodium soap from glyceryl oleate
and glyceryl palmitate. Structural formulae of these compounds
are given below.
(i)
![]() (ii)
(ii)
Answer :
 |
(i)
(ii)
![]()
Q5 :
Following type of nom-ionic detergents are present in liquid detergents, emulsifying agents and wetting agents. Label the hydrophilic and hydrophobic parts in the molecule. Identify the functional group (s) present in the molecule.
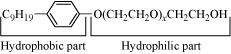 |
Answer :
Functional groups present in the molecule are:
(i) Ether, and
(ii)
primary alcoholic group
�Exercise : Solutions of Questions on Page Number : 454���������������������������������������������������������������������������������������������������������������������
Q1 :
Why do we need to classify drugs in different ways?
Answer :
The classification of drugs and the reasons for classification are as follows:
(i) On the basis of pharmacological effect:
This classification provides doctors the whole range of drugs available for the treatment of a particular type of problem. Hence, such a classification is very useful to doctors.
(ii) On the basis of drug action:
This classification is based on the action of a drug on a particular biochemical process. Thus, this classification is important.
(iii) On the basis of chemical structure:
This classification provides the range of drugs sharing common structural features and often having similar pharmacological activity.
(iv) On the basis of molecular targets:
This classification provides medicinal chemists the drugs having the same mechanism of action on targets. Hence, it is the most useful to medicinal chemists.
Q2 :
Explain the term target molecules or drug targets as used in medicinal chemistry.
Answer :
In medicinal chemistry, drug targets refer to the key molecules involved in certain metabolic pathways that result in specific diseases. Carbohydrates, proteins, lipids, and nucleic acids are examples of drug targets.
Drugs are chemical agents designed to inhibit these target molecules by binding with the active sites of the key molecules.
Q3 :
Name the macromolecules that are chosen as drug targets.
Answer :
The macromolecules chosen as drug targets are carbohydrates, lipids, proteins, and nucleic acids.
Why should not medicines be taken without consulting doctors?
Answer :
A medicine can bind to more than one receptor site. Thus, a medicine may be toxic for some receptor sites. Further, in most cases, medicines cause harmful effects when taken in higher doses than recommended. As a result, medicines may be poisonous in such cases. Hence, medicines should not be taken without consulting doctors.
Q5 :
Define the term chemotherapy.
Answer :
The use of chemicals for therapeutic effect is called chemotherapy.For example: the use of chemicals in the diagnosis, prevention, and treatment of diseases
Q6 :
Which forces are involved in holding the drugs to the active site of enzymes?
Answer :
Either of the following forces can be involved in holding drugs to the active sites of enzymes.
(i) Ionic bonding
(ii) Hydrogen bonding
(iii) Dipole - dipole interaction
(iv)
van der Waals force
Q7 :
While antacids and antiallergic drugs interfere with the function of histamines, why do these not interfere with the function of each other?
Answer :
Specific drugs affect particular receptors. Antacids and anti-allergic drugs work on different receptors. This is the reason why antacids and anti-allergic drugs do not interfere with each other's functions, but interfere with the functions of histamines.
Q8 :
Low level of noradrenaline is the cause of depression. What types of drugs are needed to cure this problem? Name two drugs.
Answer :
Anti-depressant drugs are needed to counteract the effect of depression. These drugs inhibit enzymes catalysing the degradation of the neurotransmitter, noradrenaline. As a result, the important neurotransmitter is slowly metabolised and then it can activate its receptor for longer periods of time.
Two anti-depressant drugs are:
(i) Iproniazid
(ii)
Phenelzine
Q9 :
What is meant by the term 'broad spectrum antibiotics'? Explain.
Answer :
Antibiotics that are effective against a wide range of gram-positive and gram-negative bacteria are known as broad spectrum antibiotics. Chloramphenicol is a broad spectrum antibiotic.
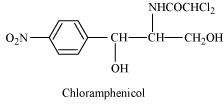 |
It can be used for the treatment of typhoid, dysentery, acute fever, pneumonia, meningitis, and certain forms of urinary infections. Two other broad spectrum antibiotics are vancomycin and ofloxacin. Ampicillin and amoxicillin - synthetically modified from penicillin - are also broad spectrum antibiotics.
![]()
Q10 :
How do antiseptics differ from disinfectants? Give one example of each.
Answer :
Antisepticsand disinfectants are effective against micro-organisms. However, antiseptics are applied to the living tissues such as wounds, cuts, ulcers, and diseased skin surfaces, while disinfectants are applied to inanimate objects such as floors, drainage system, instruments, etc. Disinfectants are harmful to the living tissues.
Iodine is an example of a strong antiseptic. Tincture of iodine (2 - 3 percent of solution of iodine in alcohol - water mixture) is applied to wounds. 1 percent solution of phenol is used as a disinfectant.
Q11 :
Why are cimetidine and ranitidine better antacids than sodium hydrogen carbonate or magnesium or aluminium hydroxide?
Answer :
Antacids such as sodium hydrogen carbonate, magnesium hydroxide, and aluminium hydroxide work by neutralising the excess hydrochloric acid present in the stomach. However, the root cause for the release of excess acid remains untreated.
Cimetidine and rantidine are better antacids as they control the root cause of acidity. These drugs prevent the interaction of histamine with the receptors present in the stomach walls. Consequently, there is a decrease in the amount of acid released by the stomach. This is why cimetidine and rantidine are better antacids than sodium hydrogen carbonate, magnesium hydroxide, and aluminium hydroxide.
Q12 :
Name a substance which can be used as an antiseptic as well as disinfectant.
Answer :
Phenol can be used as an antiseptic as well as a disinfectant. 0.2 percent solution of phenol is used as an antiseptic, while 1 per cent of its solution is used as a disinfectant.
Q13 :
What are the main constituents of dettol?
Answer :
The main constituents of dettol are chloroxylenol and α-terpineol.
 |
Q14 :
What is tincture of iodine? What is its use?
Answer :
Tincture of iodine is a 2 - 3 percent solution of iodine in alcohol - water mixture. It is applied to wounds as an antiseptic.
Q15 :
What are food preservatives?
Answer :
Food preservatives are chemicals that prevent food from spoilage due to microbial growth. Table salt, sugar, vegetable oil, sodium benzoate (C6H3COONa), and salts of propanoic acid are some examples of food preservatives.
Q16 :
Why is use of aspartame limited to cold foods and drinks?
Answer :
Aspartame becomes unstable at cooking temperature. This is the reason why its use is limited to cold foods and drinks.
Q17 :
What are artificial sweetening agents? Give two examples.
Answer :
Artificial sweetening agents are chemicals that sweeten food. However, unlike natural sweeteners, they do not add calories to our body. They do not harm the human body. Some artificial sweeteners are aspartame, saccharin, sucrolose, and alitame.
Q18 :
Name a sweetening agent used in the preparation of sweets for a diabetic patient.
Answer :
Artificial sweetening agents such as saccharin, alitame, and aspartame can be used in preparing sweets for diabetic patients.
Q19 :
What problem arises in using alitame as artificial sweetener?
Answer :
Alitame is a high potency sweetener. It is difficult to control the sweetness of food while using alitame as an artificial sweetener.
Q20 :
How are synthetic detergents better than soap?
Answer :
Soaps work in soft water. However, they are not effective in hard water. In contrast, synthetic detergents work both in soft water and hard water.
Therefore, synthetic detergents are better than soaps.
Q21 :
Explain the following terms with suitable examples
(i) Cationic detergents
(ii) Anionic detergents and
(iii) Non-ionic detergents
Answer :
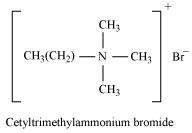
(i) Cationic detergent
Cationic detergents are quaternary ammonium salts of acetates, chlorides, or bromides. These are called cationic detergents because the cationic part of these detergents contains a long hydrocarbon chain and a positive charge on the N atom.
For example: cetyltrimethylammonium bromide
 |
(ii) Anionic detergents
Anionic detergents are of two types:
1.
3����������������������������������������������������������������������������������������������������������������������������������� 3
Sodium
alkyl sulphates: These detergents are sodium salts of
long chain alcohols. They are prepared by first
treating these alcohols with concentrated
sulphuric acid and then with sodium hydroxide. Examples
of these detergents include
sodium lauryl sulphate
(C11H23CH2OSO -Na+) and sodium
stearyl sulphate (C17H35CH2OSO -Na+).
2. Sodium alkylbenzenesulphonates: These detergents are sodium salts of long chain alkylbenzenesulphonic acids. They are prepared by Friedel-Crafts alkylation of benzene with long chain alkyl halides or alkenes. The obtained product is first treated with concentrated sulphuric acid and then with sodium hydroxide. Sodium 4-(1-dodecy) benzenesulphonate (SDS) is an example of anionic detergents.
(iii) Non-ionic detergents
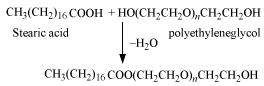 |
Molecules of these detergents do not contain any ions. These detergents are esters of alcohols having high molecular mass. They are obtained by reacting polyethylene glycol and stearic acid.
![]()
Q22 :
What are biodegradable and non-biodegradable detergents? Give one example of each.
Answer :
Detergents that can be degraded by bacteria are called biodegradable detergents. Such detergents have straight hydrocarbon chains. For example: sodium lauryl sulphate
Detergents that cannot be degraded by bacteria are called non-biodegradable detergents. Such detergents have highly-branched hydrocarbon chains. For example: sodium -4- (1, 3, 5, 7- tetra methyl octyl) benzene sulphonate
Q23 :
Why do soaps not work in hard water?
Answer :
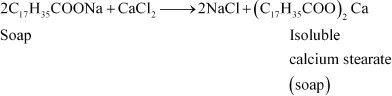 |
Soaps are sodium or potassium salts of long-chain fatty acids. Hard water contains calcium and magnesium ions. When soaps are dissolved in hard water, these ions displace sodium or potassium from their salts and form insoluble calcium or magnesium salts of fatty acids. These insoluble salts separate as scum.
This is the reason why soaps do not work in hard water.
Q24 :
Can you use soaps and synthetic detergents to check the hardness of water?
Answer :
Soaps get precipitated in hard water, but not in soft water. Therefore, soaps can be used for checking the hardness of water.
However,synthetic detergents do not get precipitated either in hard water or in soft water. Therefore, synthetic detergents cannot be used for checking the hardness of water.
Q25 :
Explain the cleansing action of soaps.
Answer :
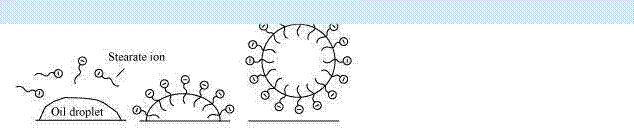 |
Soap molecules form micelles around an oil droplet (dirt) in such a way that the hydrophobic parts of the stearate ions attach themselves to the oil droplet and the hydrophilic parts project outside the oil droplet. Due to the polar nature of the hydrophilic parts, the stearate ions (along with the dirt) are pulled into water, thereby removing the dirt from the cloth.
Q26 :
If water contains dissolved calcium hydrogen carbonate, out of soaps and synthetic detergents which one will you use for cleaning clothes?
Answer :
Synthetic detergents are preferred for cleaning clothes. When soaps are dissolved in water containing calcium ions, these ions form insoluble salts that are of no further use.
However,when synthetic detergents are dissolved in water containing calcium ions, these ions form soluble salts that act as cleansing agents.
![]()
Q27 :
![]() Label the hydrophilic and hydrophobic parts in the following compounds.
Label the hydrophilic and hydrophobic parts in the following compounds.
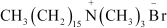 (i)
(i)
(ii)
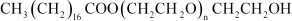 (iii)
(iii)
![]() Answer :
Answer :
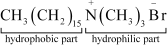 (i)
(i)
![]() (ii)
(ii)