Nuclei
NCERT Textbook Solution (Laptop/Desktop is best to view this page)
Question 13.1:
![]()
![]() Two stable
isotopes of lithiumĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀ have respective
abundances of 7.5% and 92.5%.
These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic
mass of lithium.
Two stable
isotopes of lithiumĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀ have respective
abundances of 7.5% and 92.5%.
These isotopes have masses 6.01512 u and 7.01600 u, respectively. Find the atomic
mass of lithium.
![]()
![]()
![]()
![]() Boron has two stable isotopes,ĀĀĀĀĀĀ andĀĀĀĀĀ . Their respective masses are 10.01294
u and 11.00931 u, and the atomic
mass of boron is 10.811
u. Find the abundances ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ and
Boron has two stable isotopes,ĀĀĀĀĀĀ andĀĀĀĀĀ . Their respective masses are 10.01294
u and 11.00931 u, and the atomic
mass of boron is 10.811
u. Find the abundances ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ and
.
Answer
![]()
![]()
![]()
![]() Mass of lithium isotope
, m1 = 6.01512 u Mass of lithium isotope
, m2 = 7.01600 u Abundance of , η1= 7.5%
Mass of lithium isotope
, m1 = 6.01512 u Mass of lithium isotope
, m2 = 7.01600 u Abundance of , η1= 7.5%
Abundance ofĀĀĀĀ Ā, η2= 92.5%
The atomic mass of lithium atom is given as:
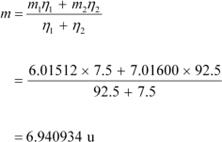 |
![]()
![]()
![]() Mass of boron isotope
, m1 = 10.01294 u Mass of boron isotope
, m2 = 11.00931 u Abundance of , η1 = x%
Mass of boron isotope
, m1 = 10.01294 u Mass of boron isotope
, m2 = 11.00931 u Abundance of , η1 = x%
![]() Abundance ofĀĀĀĀĀĀ , η2= (100 −
x)%
Abundance ofĀĀĀĀĀĀ , η2= (100 −
x)%
Atomic mass of boron, m = 10.811 u
The atomic mass of boron atom is given as:
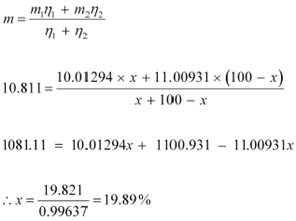 |
![]() And 100 − x = 80.11%
And 100 − x = 80.11%
![]() Hence, the abundance of
Hence, the abundance of
![]()
Question 13.2:
is 19.89% and that ofĀĀĀĀĀĀ is 80.11%.
![]()
![]() The three stable isotopes of neon:ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀĀĀ have respective abundances of 90.51%, 0.27% and 9.22%. The atomic masses of the three isotopes
are 19.99 u, 20.99 u and 21.99 u, respectively. Obtain the average atomic mass of neon.
The three stable isotopes of neon:ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀĀĀ have respective abundances of 90.51%, 0.27% and 9.22%. The atomic masses of the three isotopes
are 19.99 u, 20.99 u and 21.99 u, respectively. Obtain the average atomic mass of neon.
Answer
![]()
![]()
![]() Atomic mass ofĀĀĀĀĀĀĀĀ ,
m1= 19.99 u Abundance of , η1 = 90.51% Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m2 = 20.99 u
Atomic mass ofĀĀĀĀĀĀĀĀ ,
m1= 19.99 u Abundance of , η1 = 90.51% Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m2 = 20.99 u
![]() Abundance ofĀĀĀĀĀĀĀĀ , η2 = 0.27%
Abundance ofĀĀĀĀĀĀĀĀ , η2 = 0.27%
![]()
![]() Atomic mass ofĀĀĀĀĀĀĀĀ ,
m3 = 21.99 u Abundance of , η3 = 9.22%
Atomic mass ofĀĀĀĀĀĀĀĀ ,
m3 = 21.99 u Abundance of , η3 = 9.22%
The average atomic mass of neon is given as:
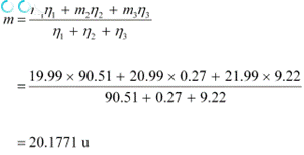 |
![]()
Question 13.3:
Obtain the binding energy (in MeV) of a nitrogen
nucleus ![]() Ā, given
Ā, given ![]()
=14.00307 u
Answer
![]() Atomic mass of nitrogenĀĀĀĀĀĀĀĀĀĀĀ , m = 14.00307
u
Atomic mass of nitrogenĀĀĀĀĀĀĀĀĀĀĀ , m = 14.00307
u
![]() A nucleus of nitrogenĀĀĀĀĀĀĀĀ contains 7 protons and 7 neutrons.
A nucleus of nitrogenĀĀĀĀĀĀĀĀ contains 7 protons and 7 neutrons.
Hence, the mass defect of this nucleus, Δm = 7mH + 7mn − m
Where,
Mass of a proton, mH = 1.007825 u Mass of a neutron, mn= 1.008665 u
∴Δm = 7 ū 1.007825 + 7 ū 1.008665 − 14.00307
= 7.054775 + 7.06055 − 14.00307
= 0.11236 u
But 1 u = 931.5 MeV/c2
∴Δm = 0.11236 ū 931.5 MeV/c2
Hence, the binding energy of the nucleus is given as:
Eb = Δmc2
Where,
c = Speed of light
∴Eb = 0.11236 ū 931.5 ![]()
= 104.66334 MeV
Hence, the binding energy of a nitrogen nucleus is 104.66334 MeV.
![]()
Question 13.4:
![]()
![]() Obtain the binding energy of the nucleiĀĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀĀĀ in units of MeV from the following data:
Obtain the binding energy of the nucleiĀĀĀĀĀĀĀĀ andĀĀĀĀĀĀĀĀĀ in units of MeV from the following data:
![]() Ā= 55.934939 u
Ā= 55.934939 u ![]() = 208.980388 u
= 208.980388 u
Answer
![]() Atomic mass ofĀĀĀĀĀĀĀ , m1 = 55.934939 u
Atomic mass ofĀĀĀĀĀĀĀ , m1 = 55.934939 u
![]() nucleus has 26 protons and (56 − 26) = 30 neutrons
nucleus has 26 protons and (56 − 26) = 30 neutrons
Hence, the mass defect of the nucleus, Δm = 26 ū mH + 30 ū mn − m1
Where,
Mass of a proton, mH = 1.007825 u Mass of a neutron, mn = 1.008665 u
∴Δm = 26 ū 1.007825 + 30 ū 1.008665 − 55.934939
= 26.20345 + 30.25995 − 55.934939
= 0.528461 u
But 1 u = 931.5 MeV/c2
∴Δm = 0.528461 ū 931.5 MeV/c2
The binding energy of this nucleus is given as:
Eb1 = Δmc2
Where,
![]() c =
Speed of light
c =
Speed of light
∴Eb1 = 0.528461 ū 931.5
= 492.26 MeV
![]()
![]()
![]() Average
binding energy per nucleon Atomic mass ofĀĀĀĀĀĀ , m2 = 208.980388 u
Average
binding energy per nucleon Atomic mass ofĀĀĀĀĀĀ , m2 = 208.980388 u
nucleus has 83 protons and (209 − 83) 126 neutrons.
Hence, the mass defect of this nucleus is given as:
Δm' = 83 ū mH + 126 ū mn − m2
Where,
Mass of a proton, mH = 1.007825 u Mass of a neutron, mn = 1.008665 u
∴Δm' = 83 ū 1.007825 + 126 ū 1.008665 − 208.980388
= 83.649475 + 127.091790 − 208.980388
= 1.760877 u
But 1 u = 931.5 MeV/c2
∴Δm' = 1.760877 ū 931.5 MeV/c2
Hence, the binding energy of this nucleus is given as:
Eb2 = Δm'c2
= 1.760877 ū 931.5 ![]()
= 1640.26 MeV
![]() Average bindingenergy per nucleon =
Average bindingenergy per nucleon =
![]()
Question 13.5:
![]() A given coin has a mass of 3.0 g. Calculate
the nuclear energy that would be required
to separate all the neutrons
and protons from each other. For simplicity
assume that the coin
A given coin has a mass of 3.0 g. Calculate
the nuclear energy that would be required
to separate all the neutrons
and protons from each other. For simplicity
assume that the coin
is entirely made ofĀĀĀĀĀĀĀĀ atoms (of mass 62.92960 u).
Answer
Mass of a copper coin, mÆ = 3 g
![]() Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀ atom, m = 62.92960
u
Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀ atom, m = 62.92960
u
![]()
![]() The total number ofĀĀĀĀĀĀĀĀĀĀĀ atoms in the coin
The total number ofĀĀĀĀĀĀĀĀĀĀĀ atoms in the coin
Where,
NA = AvogadroÆs number = 6.023 ū 1023 atoms /g
Mass number = 63 g
![]()
![]() nucleus has 29 protons and (63 − 29) 34 neutrons
nucleus has 29 protons and (63 − 29) 34 neutrons
∴Mass defect of this nucleus, Δm' = 29 ū mH + 34 ū mn − m
Where,
Mass of a proton, mH = 1.007825 u Mass of a neutron, mn = 1.008665 u
∴Δm' = 29 ū 1.007825 + 34 ū 1.008665 − 62.9296
= 0.591935 u
Mass defect of all the atoms present in the coin, Δm = 0.591935 ū 2.868 ū 1022
= 1.69766958 ū 1022 u
But 1 u = 931.5 MeV/c2
∴Δm = 1.69766958 ū 1022 ū 931.5 MeV/c2
Hence, the binding energy of the nuclei of the coin is given as:
Eb= Δmc2
= 1.69766958 ū 1022 ū 931.5 ![]()
= 1.581 ū 1025 MeV
But 1 MeV = 1.6 ū 10−13 J
Eb = 1.581 ū 1025 ū 1.6 ū 10−13
= 2.5296 ū 1012 J
This much energy is required to separate all the neutrons and protons from the given coin.
![]()
Question 13.6:
Write nuclear reaction equations for
![]()
![]() α-decay ofĀĀĀĀĀĀĀĀĀĀ (ii) α-decay of
α-decay ofĀĀĀĀĀĀĀĀĀĀ (ii) α-decay of
![]() β−-decay of
β−-decay of ![]() Ā(iv) β−-decay of
Ā(iv) β−-decay of
![]() (v) β+-decay ofĀĀĀĀĀĀĀ (vi) β+-decay
of
(v) β+-decay ofĀĀĀĀĀĀĀ (vi) β+-decay
of ![]()
(vii) Electron
capture of ![]()
Answer
![]() α is a nucleus
of heliumĀĀĀĀĀĀĀĀĀĀĀĀ and β
is an electron
(e− for β− and
e+ for β+). In every α-
α is a nucleus
of heliumĀĀĀĀĀĀĀĀĀĀĀĀ and β
is an electron
(e− for β− and
e+ for β+). In every α-
decay, there is a loss of 2 protons and 4 neutrons. In every β+-decay, there is a loss of 1 proton and a neutrino is emitted from the nucleus. In every β−-decay, there is a gain of 1 proton and an antineutrino is emitted from the nucleus.
For the given cases, the various nuclear reactions can be written as:
 |
![]()
Question 13.7:
A radioactive isotope has a half-life of T years. How long will it take the activity to reduce to a) 3.125%, b) 1% of its original value?
Answer
Half-life of the radioactive isotope = T years Original amount of the radioactive isotope = N0
After decay, the amount of the radioactive isotope = N
It is given that only 3.125% of N0 remains after decay. Hence, we can write:
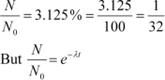 |
Where,
λ = Decay constant
t = Time
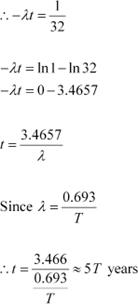 |
Hence, the isotope will take about 5T years to reduce to 3.125% of its original value.
After decay, the amount of the radioactive isotope = N
It is given that only 1% of N0 remains after decay. Hence, we can write:
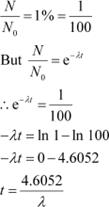 |
Since, λ = 0.693/T
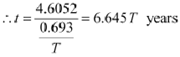 |
Hence, the isotope will take about 6.645T years to reduce to 1% of its original value.
![]()
Question 13.8:
![]()
![]() The normal activity of living carbon-containing matter is found to be about 15 decays per minute for every gram of carbon.
This activity arises from the small proportion of
The normal activity of living carbon-containing matter is found to be about 15 decays per minute for every gram of carbon.
This activity arises from the small proportion of
![]() radioactiveĀĀĀĀĀĀ present with the stable carbon isotopeĀĀĀĀĀĀ . When the organism is dead, its interaction with the atmosphere
(which maintains the above equilibrium activity)
radioactiveĀĀĀĀĀĀ present with the stable carbon isotopeĀĀĀĀĀĀ . When the organism is dead, its interaction with the atmosphere
(which maintains the above equilibrium activity)
![]() ceases and its activity
begins to drop. From the known half-life
(5730 years) ofĀĀĀĀĀĀ , and the measured
activity, the age of the specimen can be approximately estimated. This is the
ceases and its activity
begins to drop. From the known half-life
(5730 years) ofĀĀĀĀĀĀ , and the measured
activity, the age of the specimen can be approximately estimated. This is the
principle ofĀĀĀĀĀĀ dating used in archaeology. Suppose a specimen from Mohenjodaro gives an activity of 9 decays per minute per gram of carbon. Estimate the approximate age of the Indus-Valley civilisation.
Answer
Decay rate of living carbon-containing matter, R = 15 decay/min
Let N be the number of radioactive atoms present in a normal carbon- containing matter.
![]()
![]() Half life ofĀĀĀĀĀ ,ĀĀĀĀĀĀ = 5730 years
Half life ofĀĀĀĀĀ ,ĀĀĀĀĀĀ = 5730 years
The decay rate of the specimen obtained from the Mohenjodaro site:
R' = 9 decays/min
Let N' be the number of radioactive atoms present in the specimen during the Mohenjodaro period.
Therefore, we can relate the decay constant, λand time, t as:
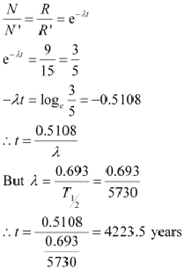 |
Hence, the approximate age of the Indus-Valley civilisation is 4223.5 years.
![]()
Question 13.9:
![]()
![]() Obtain the amount ofĀĀĀĀĀĀĀĀ necessary to provide a radioactive source of 8.0 mCi strength.
The half-life ofĀ is 5.3 years.
Obtain the amount ofĀĀĀĀĀĀĀĀ necessary to provide a radioactive source of 8.0 mCi strength.
The half-life ofĀ is 5.3 years.
Answer
The strength of the radioactive source is given as:
 |
Where,
N = Required number of atoms
![]()
![]() Half-life ofĀĀĀĀĀĀĀ ,ĀĀĀĀĀĀ = 5.3 years
Half-life ofĀĀĀĀĀĀĀ ,ĀĀĀĀĀĀ = 5.3 years
= 5.3 ū 365 ū 24 ū 60 ū 60
= 1.67 ū 108 s
For decay constant λ, we have the rate of decay as:
Where, λ ![]()
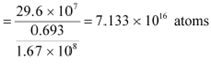 |
ForĀĀĀĀĀĀĀĀĀĀ :
![]()
![]() Mass of 6.023 ū 1023 (AvogadroÆs number) atoms = 60 g
Mass of 6.023 ū 1023 (AvogadroÆs number) atoms = 60 g
![]() ∴Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ atoms
∴Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ atoms
![]() Hence, the amount of
Hence, the amount of
![]()
Question 13.10:
necessary for the purpose is 7.106 ū 10−6 g.
![]() The half-life
ofĀĀĀĀĀĀĀ is 28 years. What is the disintegration rate of 15 mg of this isotope?
The half-life
ofĀĀĀĀĀĀĀ is 28 years. What is the disintegration rate of 15 mg of this isotope?
Answer
![]()
![]() Half life ofĀĀĀĀĀĀĀ ,ĀĀĀĀĀ = 28 years
Half life ofĀĀĀĀĀĀĀ ,ĀĀĀĀĀ = 28 years
= 28 ū 365 ū 24 ū 60 ū 60
= 8.83 ū 108 s
Mass of the isotope, m = 15 mg
Rate of disintegration, Ā![]() ĀWhere,
ĀWhere,
λ = Decay constant ![]()
Hence, the disintegration rate of 15 mg of the given isotope is 7.878 ū 1010 atoms/s.
![]()
Question 13.11:
![]()
![]() Obtain approximately the ratio of the nuclear radii of the gold isotope silver isotopeĀĀĀĀĀĀĀĀĀĀ .
Obtain approximately the ratio of the nuclear radii of the gold isotope silver isotopeĀĀĀĀĀĀĀĀĀĀ .
Answer
and the
![]() Nuclear radius of the gold isotopeĀĀĀĀĀĀĀĀĀĀĀĀĀ = RAu
Nuclear radius of the gold isotopeĀĀĀĀĀĀĀĀĀĀĀĀĀ = RAu
![]() Nuclear radius
of the silver isotopeĀĀĀĀĀĀĀĀĀĀĀĀĀ = RAg
Nuclear radius
of the silver isotopeĀĀĀĀĀĀĀĀĀĀĀĀĀ = RAg
Mass number of gold, AAu = 197 Mass number of silver, AAg = 107
The ratio of the radii of the two nuclei is related with their mass numbers as:
 |
Hence, the ratio of the nuclear radii of the gold and silver isotopes is about 1.23.
![]()
Question 13.12:
![]()
![]()
![]()
![]() Find the Q-value and the kinetic
energy of the emitted α-particle in the α-decay of (a) and (b)ĀĀĀĀĀĀĀ .
Find the Q-value and the kinetic
energy of the emitted α-particle in the α-decay of (a) and (b)ĀĀĀĀĀĀĀ .
GivenĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 226.02540 u,ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 222.01750 u,
![]() Ā= 220.01137 u,
Ā= 220.01137 u, ![]() = 216.00189 u.
= 216.00189 u.
Answer
![]() Alpha particle
decay ofĀĀĀĀĀĀĀĀĀĀ emits a
helium nucleus. As a result, its mass number
reduces to (226 − 4) 222 and its atomic
number reduces to (88 −
2) 86. This is shown in the following nuclear
reaction.
Alpha particle
decay ofĀĀĀĀĀĀĀĀĀĀ emits a
helium nucleus. As a result, its mass number
reduces to (226 − 4) 222 and its atomic
number reduces to (88 −
2) 86. This is shown in the following nuclear
reaction.
Q-value of
emitted α-particle = (Sum of initial mass − Sum of final mass) c2
Where,
 |
c = Speed of light It is given that:
Q-value = [226.02540 − (222.01750 + 4.002603)] u c2
= 0.005297 u c2
But 1 u = 931.5 MeV/c2
∴Q = 0.005297 ū 931.5 ≈ 4.94 MeV
![]() Kinetic energy of the α-particle
Kinetic energy of the α-particle ![]()
Alpha particle decay ofĀĀĀĀĀĀĀĀĀĀĀĀĀ is shown by the following nuclear reaction.
It is given that:
![]() Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀ = 220.01137 u
Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀ = 220.01137 u
![]() Mass ofĀĀĀĀĀĀĀĀĀĀĀĀ = 216.00189 u
Mass ofĀĀĀĀĀĀĀĀĀĀĀĀ = 216.00189 u
∴Q-value = ![]()
![]() ≈ 641 MeV
≈ 641 MeV
Kinetic energy of the α-particle
= 6.29 MeV
![]()
Question 13.13:
The radionuclide 11C decays according to
The maximum energy of the emitted positron is 0.960 MeV.
Given the mass values:
calculate Q and compare it with the maximum energy of the positron emitted
Answer
The given nuclear reaction is:
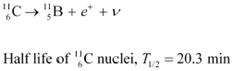 |
![]() Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀ = 11.011434
u Atomic mass of
Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀ = 11.011434
u Atomic mass of ![]()
Maximum energy possessed by the emitted positron = 0.960 MeV
![]() The change in the Q-value (ΔQ) of the nuclear
masses of theĀĀĀĀĀĀ nucleus is given as:
The change in the Q-value (ΔQ) of the nuclear
masses of theĀĀĀĀĀĀ nucleus is given as:
Where,
me = Mass of an electron or positron = 0.000548 u
c = Speed of light
mÆ = Respective nuclear masses
If atomic masses are used instead of nuclear masses, then we have to add 6 me in the case
![]()
![]() ofĀĀĀĀĀ and 5 me in the case ofĀĀĀĀĀ .
ofĀĀĀĀĀ and 5 me in the case ofĀĀĀĀĀ .
Hence, equation (1) reduces to:
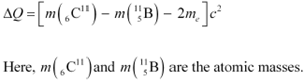 |
∴ΔQ = [11.011434 − 11.009305 − 2 ū 0.000548] c2
= (0.001033 c2) u
But 1 u = 931.5 Mev/c2
∴ΔQ = 0.001033 ū 931.5 ≈ 0.962 MeV
The value of Q is almost comparable to the maximum energy of the emitted positron.
![]()
Question 13.14:
![]()
![]()
![]() The nucleusĀĀĀĀĀĀĀĀ decays byĀĀĀĀĀ emission. Write down theĀĀĀĀ decay equation and
The nucleusĀĀĀĀĀĀĀĀ decays byĀĀĀĀĀ emission. Write down theĀĀĀĀ decay equation and
determine the maximum kinetic energy of the electrons emitted. Given that:
![]() = 22.994466
u
= 22.994466
u
![]() = 22.989770
u.
= 22.989770
u.
Answer
![]() InĀĀĀĀĀĀ emission, the number of protons increases
by 1, and one electron
and an antineutrino are emitted
from the parent nucleus.
InĀĀĀĀĀĀ emission, the number of protons increases
by 1, and one electron
and an antineutrino are emitted
from the parent nucleus.
![]()
![]() emission of the nucleusĀĀĀĀĀĀĀĀĀ is given as:
emission of the nucleusĀĀĀĀĀĀĀĀĀ is given as:
It is given that:
![]() Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 22.994466
u
Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 22.994466
u
![]() Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 22.989770
u
Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 22.989770
u
Mass of an electron, me = 0.000548 u
Q-value of the given reaction is given as:
![]()
![]() There are 10 electrons
inĀĀĀĀĀĀĀĀĀ and 11 electrons
inĀĀĀĀĀĀĀĀ . Hence, the mass of the electron
There are 10 electrons
inĀĀĀĀĀĀĀĀĀ and 11 electrons
inĀĀĀĀĀĀĀĀ . Hence, the mass of the electron
is cancelled in the Q-value equation.
 |
![]()
![]() The daughter
nucleus is too heavy as compared toĀĀĀĀĀ andĀĀĀ . Hence, it carries negligible energy. The kinetic
energy of the antineutrino is nearly zero. Hence, the maximum kinetic
energy of the emitted electrons
is almost equal to the Q-value, i.e., 4.374 MeV.
The daughter
nucleus is too heavy as compared toĀĀĀĀĀ andĀĀĀ . Hence, it carries negligible energy. The kinetic
energy of the antineutrino is nearly zero. Hence, the maximum kinetic
energy of the emitted electrons
is almost equal to the Q-value, i.e., 4.374 MeV.
![]()
Question 13.15:
The Q value of a nuclear reaction A + b → C + d is defined by
Q = [ mA+ mb− mC− md]c2 where the masses refer to the respective nuclei. Determine from the given data the Q-value of the following reactions and state whether the reactions are exothermic or endothermic.
Atomic masses are given to be
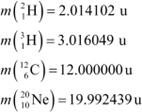 |
Answer
The given nuclear reaction is:
It is given that:
![]() Atomic mass
Atomic mass
![]()
![]() Atomic mass Atomic mass
Atomic mass Atomic mass
According to the question, the Q-value of the reaction can be written as:
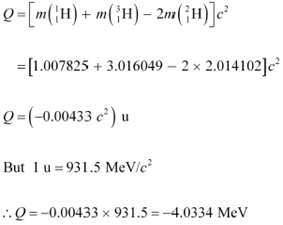
The negativeQ-value of the reaction shows that the reaction is endothermic.
The given nuclear reaction is:
It is given that:
Atomic mass of Ā![]() ĀĀAtomic mass of Ā
ĀĀAtomic mass of Ā![]() ĀAtomic mass of Ā
ĀAtomic mass of Ā![]() ĀThe Q-value of this reaction
is given as:
ĀThe Q-value of this reaction
is given as:
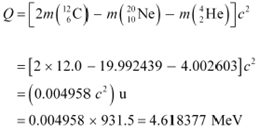 |
The positive Q-value of the reaction shows that the reaction is exothermic.
![]()
Question 13.16:
gue b ive
![]() Suppose, we think of fission of aĀĀĀĀĀĀĀĀ nucleus into two equal fragments, fission
energetically possible? Ar y working out Q of the process.
G
Suppose, we think of fission of aĀĀĀĀĀĀĀĀ nucleus into two equal fragments, fission
energetically possible? Ar y working out Q of the process.
G
. Is the
![]()
![]() n
n
![]()
![]() andĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ .
andĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ .
Answer
![]() The fission ofĀĀĀĀĀĀĀ can be given as:
The fission ofĀĀĀĀĀĀĀ can be given as:
It is given that:
![]() Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 55.93494
u Atomic mass of
Atomic mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 55.93494
u Atomic mass of ![]()
The Q-value of this nuclear reaction is given as:
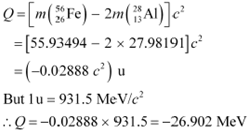 |
The Q-value of the fission is negative. Therefore, the fission is not possible energetically. For an energetically-possible fission reaction, the Q-value must be positive.
![]()
Question 13.17:
![]()
![]() The fission properties ofĀĀĀĀĀĀĀĀĀ are very similar to those ofĀĀĀĀĀĀĀ .
The fission properties ofĀĀĀĀĀĀĀĀĀ are very similar to those ofĀĀĀĀĀĀĀ .
![]() The average energy released
per fission is 180 MeV. How much energy, in MeV, is released if all the atoms in 1 kg of pureĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ undergo fission?
The average energy released
per fission is 180 MeV. How much energy, in MeV, is released if all the atoms in 1 kg of pureĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ undergo fission?
Answer
![]()
![]() Average energy released per fission ofĀĀĀĀĀĀĀĀĀ ,
Average energy released per fission ofĀĀĀĀĀĀĀĀĀ ,
![]()
![]() Amount of pureĀĀĀĀĀĀĀĀĀĀ , m = 1 kg = 1000 g NA= Avogadro
number = 6.023 ū 1023
Amount of pureĀĀĀĀĀĀĀĀĀĀ , m = 1 kg = 1000 g NA= Avogadro
number = 6.023 ū 1023
Mass number ofĀĀĀĀĀĀĀĀ = 239 g
![]() 1 mole ofĀĀĀĀĀĀĀĀĀĀĀ contains NA atoms.
1 mole ofĀĀĀĀĀĀĀĀĀĀĀ contains NA atoms.
![]()
![]() ∴m g ofĀĀĀĀĀĀĀĀĀĀĀ contains
∴m g ofĀĀĀĀĀĀĀĀĀĀĀ contains
![]() ∴Total energy released during the fission
of 1 kg ofĀĀĀĀĀĀĀĀĀ is calculated as:
∴Total energy released during the fission
of 1 kg ofĀĀĀĀĀĀĀĀĀ is calculated as:
![]()
![]() Hence,ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ is released
if all the atoms in 1 kg of pure fission.
Hence,ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ is released
if all the atoms in 1 kg of pure fission.
![]()
Question 13.18:
undergo
![]() A 1000 MW fission reactor consumes
half of its fuel in 5.00 y. How muchĀĀĀĀĀĀĀĀ did it
A 1000 MW fission reactor consumes
half of its fuel in 5.00 y. How muchĀĀĀĀĀĀĀĀ did it
![]() contain initially?
Assume that the reactor operates
80% of the time, that all the energy
contain initially?
Assume that the reactor operates
80% of the time, that all the energy
generated arises from the fission ofĀĀĀĀĀĀĀĀ and that this nuclide is consumed only by the fission process.
Answer
Half life of the fuel of the fission reactor, ![]() Āyears
Āyears
= 5 ū 365 ū 24 ū 60 ū 60 s
![]() We know that in the fission of 1 g ofĀĀĀĀĀĀĀĀ nucleus, the energy released
is equal to 200 MeV.
We know that in the fission of 1 g ofĀĀĀĀĀĀĀĀ nucleus, the energy released
is equal to 200 MeV.
1 mole, i.e., 235 g of ![]() Ācontains
6.023 ū 1023 atoms.
Ācontains
6.023 ū 1023 atoms.
![]() ∴1 gĀĀĀĀĀĀĀĀĀ contains
∴1 gĀĀĀĀĀĀĀĀĀ contains ![]()
![]() The total energy generated
per gram ofĀĀĀĀĀĀĀ is calculated as:
The total energy generated
per gram ofĀĀĀĀĀĀĀ is calculated as:
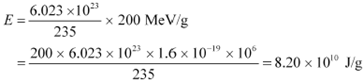 |
The reactor operates only 80% of the time.
![]() Hence, the amount ofĀĀĀĀĀĀĀĀ consumed in 5 years by the 1000 MW fission reactor is calculated as:
Hence, the amount ofĀĀĀĀĀĀĀĀ consumed in 5 years by the 1000 MW fission reactor is calculated as:
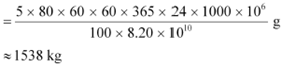 |
![]() ∴Initial amount ofĀĀĀĀĀĀĀĀ = 2 ū 1538 = 3076 kg
∴Initial amount ofĀĀĀĀĀĀĀĀ = 2 ū 1538 = 3076 kg
![]()
Question 13.19:
How long can an electric lamp of 100W be kept glowing by fusion of 2.0 kg of deuterium? Take the fusion reaction as
Answer
The given fusion reaction is:
Amount of deuterium, m = 2 kg
![]() 1 mole,
i.e., 2 g of deuterium
contains 6.023 ū 1023 atoms.
1 mole,
i.e., 2 g of deuterium
contains 6.023 ū 1023 atoms.
∴2.0 kg of deuterium contains
It can be inferred from the given reaction that when two atoms of deuterium fuse, 3.27 MeV energy is released.
∴Total energy per nucleus released in the fusion reaction:
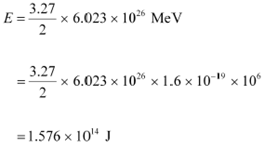 |
Power of the electric lamp, P = 100 W = 100 J/s
Hence, the energy consumed by the lamp per second = 100 J
The total time for which the electric lamp will glow is calculated as:
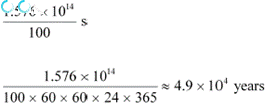 |
![]()
Question 13.20:
Calculate the height of the potential barrier for a head on collision of two deuterons. (Hint: The height of the potential barrier is given by the Coulomb repulsion between the two deuterons when they just touch each other. Assume that they can be taken as hard spheres of radius 2.0 fm.)
Answer
When two deuterons collide head-on, the distance between their centres, d is given as: Radius of 1st deuteron + Radius of 2nd deuteron
Radius of a deuteron nucleus = 2 fm = 2 ū 10−15 m
∴d = 2 ū 10−15 + 2 ū 10−15 = 4 ū 10−15 m
Charge on a deuteron nucleus = Charge on an electron = e = 1.6 ū 10−19 C Potential energy of the two-deuteron system:
Where,
![]() = Permittivity of free space
= Permittivity of free space
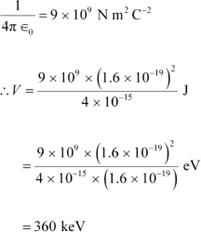 |
Hence, the height of the potential barrier of the two-deuteron system is
360 keV.
![]()
Question 13.21:
From the relation R = R0A1/3, where R0 is a constant and A is the mass number of a nucleus, show that the nuclear matter density is nearly constant (i.e. independent of A).
Answer
We have the expression for nuclear radius as:
R = R0A1/3
Where,
R0 = Constant.
A = Mass number of the nucleus
Nuclear matter density,
![]() ĀLet m be the average mass of the nucleus.
ĀLet m be the average mass of the nucleus.
Hence, mass of the nucleus = mA
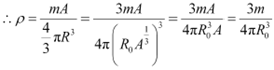 |
Hence, the nuclear matter density is independent of A. It is nearly constant.
![]()
Question 13.22:
![]() For theĀĀĀĀĀĀ (positron) emission
from a nucleus, there is another competing
process known as electron capture (electron from an
inner orbit, say, the K−shell, is captured by the nucleus and a neutrino is emitted).
For theĀĀĀĀĀĀ (positron) emission
from a nucleus, there is another competing
process known as electron capture (electron from an
inner orbit, say, the K−shell, is captured by the nucleus and a neutrino is emitted).
![]() Show that ifĀĀĀĀĀĀ emission is energetically allowed,
electron capture is necessarily allowed
but not vice−versa.
Show that ifĀĀĀĀĀĀ emission is energetically allowed,
electron capture is necessarily allowed
but not vice−versa.
Answer
Let the amount of energy released during the electron capture process be Q1. The nuclear reaction can be written as:
Let the amount of energy released during the positron capture process be Q2. The nuclear reaction can be written as:
![]()
![]() = Nuclear
mass of
= Nuclear
mass of
![]()
![]() = Nuclear
mass of
= Nuclear
mass of
![]()
![]() = Atomic
mass of
= Atomic
mass of
![]()
![]() = Atomic
mass of
= Atomic
mass of
me = Mass of an electron
c = Speed of light
Q-value of the electron capture reaction is given as:
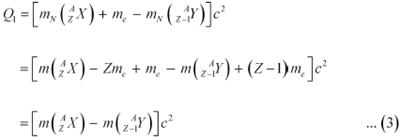
Q-value of the positron capture reaction is given as:
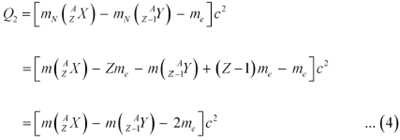 |
It can be inferred that if Q2 > 0, then Q1 > 0; Also, if Q1> 0, it does not necessarily mean that Q2 > 0.
![]() In other words, this means that ifĀĀĀĀĀ emission is energetically allowed,
then the electron capture process is necessarily allowed, but not vice-versa. This is because the Q-value must be positive
for an energetically-allowed nuclear reaction.
In other words, this means that ifĀĀĀĀĀ emission is energetically allowed,
then the electron capture process is necessarily allowed, but not vice-versa. This is because the Q-value must be positive
for an energetically-allowed nuclear reaction.
![]()
Question 13.23:
![]()
![]()
![]() In a periodic table the average
atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance
on earth. The three isotopes
In a periodic table the average
atomic mass of magnesium is given as 24.312 u. The average value is based on their relative natural abundance
on earth. The three isotopes
![]() and their masses areĀĀĀĀĀĀĀĀĀ (23.98504u),ĀĀĀĀĀĀĀĀĀĀ (24.98584u) andĀĀĀĀĀĀĀĀĀĀ (25.98259u). The
and their masses areĀĀĀĀĀĀĀĀĀ (23.98504u),ĀĀĀĀĀĀĀĀĀĀ (24.98584u) andĀĀĀĀĀĀĀĀĀĀ (25.98259u). The
natural abundance ofĀĀĀĀĀĀĀĀĀ is 78.99% by mass. Calculate the abundances of other two isotopes.
Answer
Average atomic mass of magnesium, m = 24.312 u
![]() Mass of magnesium isotopeĀĀĀĀĀĀĀĀ , m1 = 23.98504
u
Mass of magnesium isotopeĀĀĀĀĀĀĀĀ , m1 = 23.98504
u
Mass of magnesium isotope Mass of magnesium isotope
, m2 = 24.98584 u
![]()
![]() , m3 = 25.98259 u
, m3 = 25.98259 u
![]()
![]()
![]() Abundance ofĀĀĀĀĀĀĀĀ , η1= 78.99% Abundance ofĀĀ , η2 = x%
Abundance ofĀĀĀĀĀĀĀĀ , η1= 78.99% Abundance ofĀĀ , η2 = x%
Hence, abundance ofĀĀĀĀĀĀĀĀ , η3 = 100 − x − 78.99% = (21.01 − x)%
We have the relation for the average atomic mass as:
 |
![]() Hence, the abundance of
Hence, the abundance of
![]()
Question 13.24:
is 9.3% and that ofĀĀĀĀĀĀĀĀĀĀ is 11.71%.
![]()
![]() The neutron separation energy is defined as the energy required to remove a neutron from
The neutron separation energy is defined as the energy required to remove a neutron from
![]() the nucleus.
Obtain the neutron separation energies
of the nucleiĀĀĀĀĀĀĀĀĀ and following data:
the nucleus.
Obtain the neutron separation energies
of the nucleiĀĀĀĀĀĀĀĀĀ and following data:
![]() = 39.962591
u
= 39.962591
u
![]() ) = 40.962278 u
) = 40.962278 u
![]() = 25.986895
u
= 25.986895
u
![]() ) = 26.981541 u
) = 26.981541 u
Answer
from the
![]()
![]() For For
For For
![]()
![]() A neutronĀĀĀĀĀĀĀĀĀ is removed from aĀĀĀĀĀĀĀĀĀ nucleus. The corresponding nuclear
reaction can be written as:
A neutronĀĀĀĀĀĀĀĀĀ is removed from aĀĀĀĀĀĀĀĀĀ nucleus. The corresponding nuclear
reaction can be written as:
It is given that:
![]() MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 39.962591 u
MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 39.962591 u
![]() MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀ ) = 40.962278 u
MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀ ) = 40.962278 u
![]() MassĀĀĀĀĀĀĀĀĀĀĀĀĀ = 1.008665
u
MassĀĀĀĀĀĀĀĀĀĀĀĀĀ = 1.008665
u
The mass defect of this reaction is given as:
![]() Δm =
Δm =
∴Δm = 0.008978 ū 931.5 MeV/c2
Hence, the energy required for neutron removal is calculated as:
![]() ForĀĀĀĀĀĀĀ , the neutron removal
reaction can be written as:
ForĀĀĀĀĀĀĀ , the neutron removal
reaction can be written as:
It is given that:
![]() MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 26.981541
u
MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 26.981541
u
![]() MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 25.986895
u
MassĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 25.986895
u
The mass defect of this reaction is given as:
 |
Hence, the energy required for neutron removal is calculated as:
![]()
Question 13.25:
![]()
![]() A source
contains two phosphorous radio nuclidesĀĀĀĀĀĀ (T1/2 = 14.3d) andĀĀĀĀĀĀ (T1/2 =
A source
contains two phosphorous radio nuclidesĀĀĀĀĀĀ (T1/2 = 14.3d) andĀĀĀĀĀĀ (T1/2 =
![]() 25.3d). Initially, 10% of the decays come fromĀĀĀĀĀ . How long one must wait until 90% do so?
25.3d). Initially, 10% of the decays come fromĀĀĀĀĀ . How long one must wait until 90% do so?
Answer
![]()
![]()
![]() Half life ofĀĀĀĀĀ , T1/2 = 14.3 days Half life ofĀĀĀĀĀĀĀĀĀ , TÆ1/2 = 25.3 days
Half life ofĀĀĀĀĀ , T1/2 = 14.3 days Half life ofĀĀĀĀĀĀĀĀĀ , TÆ1/2 = 25.3 days
nucleus decay is 10% of the total amount of decay.
![]()
![]() The source has initially
10% ofĀĀĀĀĀĀ nucleus and 90% ofĀĀĀĀĀĀ nucleus.
The source has initially
10% ofĀĀĀĀĀĀ nucleus and 90% ofĀĀĀĀĀĀ nucleus.
![]()
![]() Suppose after t days,
the source has 10% ofĀĀĀĀĀĀ nucleus and 90% ofĀĀĀĀĀĀ nucleus.
Suppose after t days,
the source has 10% ofĀĀĀĀĀĀ nucleus and 90% ofĀĀĀĀĀĀ nucleus.
Initially:
![]() Number ofĀĀĀĀĀĀ nucleus = N
Number ofĀĀĀĀĀĀ nucleus = N
![]() Number ofĀĀĀĀĀĀ nucleus = 9 N
Number ofĀĀĀĀĀĀ nucleus = 9 N
Finally:
![]()
![]()
![]() Number of Number of
Number of Number of
ForĀĀĀĀĀĀĀ nucleus, we can write the number ratio as:
 |
![]() ForĀĀĀĀĀĀ , we can write the number ratio as:
ForĀĀĀĀĀĀ , we can write the number ratio as:
 |
On dividing equation (1) by equation (2), we get:
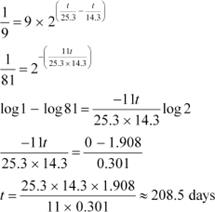 |
![]() Hence, it will take about 208.5 days for 90% decay ofĀĀĀĀĀĀĀĀ .
Hence, it will take about 208.5 days for 90% decay ofĀĀĀĀĀĀĀĀ .
![]()
Question 13.26:
Under certain circumstances, a nucleus can decay by emitting a particle more massive
than an α-particle. Consider the following decay processes:
Calculate the Q-values for these decays and determine that both are energetically allowed.
Answer
![]() Take aĀĀĀĀĀĀ emission nuclear reaction:
Take aĀĀĀĀĀĀ emission nuclear reaction:
We know that:
![]() Mass ofĀĀĀĀĀĀĀĀĀĀ m1 = 223.01850 u
Mass ofĀĀĀĀĀĀĀĀĀĀ m1 = 223.01850 u
![]()
![]() Mass ofĀĀĀĀĀĀĀĀĀĀ m2 = 208.98107 u Mass of , m3 = 14.00324 u
Mass ofĀĀĀĀĀĀĀĀĀĀ m2 = 208.98107 u Mass of , m3 = 14.00324 u
Hence, the Q-value of the reaction is given as:
Q = (m1 − m2 − m3) c2
= (223.01850 − 208.98107 − 14.00324) c2
= (0.03419 c2) u
But 1 u = 931.5 MeV/c2
∴Q = 0.03419 ū 931.5
= 31.848 MeV
Hence, the Q-value of the nuclear reaction is 31.848 MeV. Since the value is positive, the reaction is energetically allowed.
![]() Now take aĀĀĀĀĀĀĀĀ emission nuclear reaction:
Now take aĀĀĀĀĀĀĀĀ emission nuclear reaction:
We know that:
![]() Mass ofĀĀĀĀĀĀĀĀĀĀ m1 = 223.01850
Mass ofĀĀĀĀĀĀĀĀĀĀ m1 = 223.01850
![]()
![]() Mass ofĀĀĀĀĀĀĀĀĀĀ m2 =
219.00948 Mass ofĀĀĀ ,
m3 = 4.00260
Mass ofĀĀĀĀĀĀĀĀĀĀ m2 =
219.00948 Mass ofĀĀĀ ,
m3 = 4.00260
Q-value of this nuclear reaction is given as:
Q = (m1 − m2 − m3) c2
= (223.01850 − 219.00948 − 4.00260) C2
= (0.00642 c2) u
= 0.00642 ū 931.5 = 5.98 MeV
Hence, the Q value of the second nuclear reaction is 5.98 MeV. Since the value is positive, the reaction is energetically allowed.
![]()
Question 13.27:
![]() Consider the fission ofĀĀĀĀĀĀĀĀ by fast neutrons. In one fission event, no neutrons are
Consider the fission ofĀĀĀĀĀĀĀĀ by fast neutrons. In one fission event, no neutrons are
![]()
![]() emitted and the final end products,
after the beta decay of the primary fragments, are
emitted and the final end products,
after the beta decay of the primary fragments, are
andĀĀĀĀĀĀĀĀ . Calculate Q for this fission process. The relevant atomic and particle masses are
![]() mĀĀĀĀĀĀĀĀĀĀĀĀ =238.05079 u
mĀĀĀĀĀĀĀĀĀĀĀĀ =238.05079 u
![]() mĀĀĀĀĀĀĀĀĀĀĀĀĀ =139.90543 u
mĀĀĀĀĀĀĀĀĀĀĀĀĀ =139.90543 u
![]() mĀĀĀĀĀĀĀĀĀĀĀĀ = 98.90594
u
mĀĀĀĀĀĀĀĀĀĀĀĀ = 98.90594
u
Answer
![]() In the fission ofĀĀĀĀĀĀĀ , 10 β− particles
decay from the parent nucleus.
The nuclear reaction
can be written as:
In the fission ofĀĀĀĀĀĀĀ , 10 β− particles
decay from the parent nucleus.
The nuclear reaction
can be written as:
It is given that:
![]()
![]()
![]()
![]() Mass of a nucleusĀĀĀĀĀĀĀ m1 =
238.05079 u Mass of a nucleusĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ m2 = 139.90543
u Mass of a nucleusĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m3 = 98.90594 u
Mass of a nucleusĀĀĀĀĀĀĀ m1 =
238.05079 u Mass of a nucleusĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ m2 = 139.90543
u Mass of a nucleusĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m3 = 98.90594 u
Mass of a neutronĀĀĀĀĀĀ m4 = 1.008665 u
Q-value of the above equation,
Where,
mÆ = Represents the corresponding atomic masses of the nuclei
![]() = m1 −
92me
= m1 −
92me
![]() = m2 −
58me
= m2 −
58me
![]() = m3 −
44me
= m3 −
44me
![]() = m4
= m4
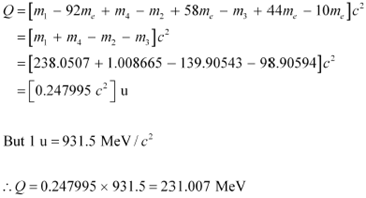
Hence, the Q-value of the fission process is 231.007 MeV.
![]()
Question 13.28:
Consider the D−T reaction (deuterium−tritium fusion)
Calculate the energy released in MeV in this reaction from the data:
![]() = 2.014102
u
= 2.014102
u
![]() = 3.016049
u
= 3.016049
u
(b)Consider the radius of both deuterium and tritium to be approximately 2.0 fm. What is the kinetic energy needed to overcome the coulomb repulsion between the two nuclei? To what temperature must the gas be heated to initiate the reaction? (Hint: Kinetic energy required for one fusion event =average thermal kinetic energy available with the interacting particles = 2(3kT/2); k = BoltzmanÆs constant, T = absolute temperature.)
Answer
![]() Take the D-T nuclear reaction:
Take the D-T nuclear reaction:
It is given that:
![]()
![]()
![]()
![]() Mass ofĀĀĀĀĀ , m1= 2.014102 u Mass ofĀĀĀĀĀ ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m2 = 3.016049 u Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ m3 = 4.002603 u
Mass ofĀĀĀĀĀ , m1= 2.014102 u Mass ofĀĀĀĀĀ ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ , m2 = 3.016049 u Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ ĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ m3 = 4.002603 u
Mass ofĀĀĀĀ , m4 = 1.008665 u
Q-value of the given D-T reaction is:
Q = [m1 + m2− m3 − m4] c2
= [2.014102 + 3.016049 − 4.002603 − 1.008665] c2
= [0.018883 c2] u
But 1 u = 931.5 MeV/c2
∴Q = 0.018883 ū 931.5 = 17.59 MeV
Radius of deuterium and tritium, r ≈ 2.0 fm = 2 ū 10−15 m
Distance between the two nuclei at the moment when they touch each other, d = r + r = 4
ū 10−15 m
Charge on the deuterium nucleus = e
Charge on the tritium nucleus = e
Hence, the repulsive potential energy between the two nuclei is given as:
Where,
∈0 = Permittivity of free space
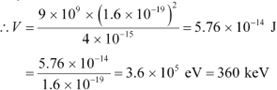 |
Hence, 5.76 ū 10−14 J or
of kinetic energy (KE) is needed to overcome the
![]() Coulomb repulsion
between the two nuclei.
Coulomb repulsion
between the two nuclei.
However, it is given that:
![]() KE
KE
Where,
k = Boltzmann constant = 1.38 ū 10−23 m2 kg s−2 K−1
T = Temperature required for triggering the reaction
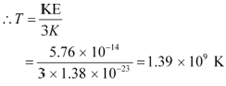 |
Hence, the gas must be heated to a temperature of 1.39 ū 109 K to initiate the reaction.
![]()
Question 13.29:
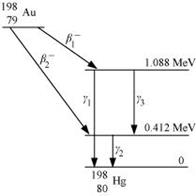
Obtain the maximum kinetic energy of β-particles, and the radiation frequencies of γ
decays in the decay scheme shown in Fig. 13.6. You are given that
m (198Au) = 197.968233 u
m (198Hg) =197.966760 u
 |
Answer
It can be observed from the given γ-decay diagram that γ1 decays from the 1.088 MeV energy level to the 0 MeV energy level.
Hence, the energy corresponding to γ1-decay is given as:
E1 = 1.088 − 0 = 1.088 MeV
hν1= 1.088 ū 1.6 ū 10−19 ū 106 J
Where,
h = PlanckÆs constant = 6.6 ū 10−34 Js
ν1 = Frequency of radiation radiated by γ1-decay
It can be observed from the given γ-decay diagram that γ2 decays from the 0.412 MeV energy level to the 0 MeV energy level.
Hence, the energy corresponding to γ2-decay is given as:
E2 = 0.412 − 0 = 0.412 MeV
hν2= 0.412 ū 1.6 ū 10−19 ū 106 J
Where,
ν2 = Frequency of radiation radiated by γ2-decay
It can be observed from the given γ-decay diagram that γ3 decays from the 1.088 MeV energy level to the 0.412 MeV energy level.
Hence, the energy corresponding to γ3-decay is given as:
E3 = 1.088 − 0.412 = 0.676 MeV
hν3= 0.676 ū 10−19 ū 106 J
Where,
ν3 = Frequency of radiation radiated by γ3-decay
![]() Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 197.968233 u
Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 197.968233 u
![]() Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 197.966760 u 1 u = 931.5 MeV/c2
Mass ofĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀĀ = 197.966760 u 1 u = 931.5 MeV/c2
Energy of the highest level is given as:
β1 decays from the 1.3720995 MeV level to the 1.088 MeV level
∴Maximum kinetic energy of the β1 particle = 1.3720995 − 1.088
= 0.2840995 MeV
β2 decays from the 1.3720995 MeV level to the 0.412 MeV level
∴Maximum kinetic energy of the β2 particle = 1.3720995 − 0.412
= 0.9600995 MeV
![]()
Question 13.30:
Calculate and compare the energy released by a) fusion of 1.0 kg of hydrogen deep within Sun and b) the fission of 1.0 kg of 235U in a fission reactor.
Answer
Amount of hydrogen, m = 1 kg = 1000 g
1 mole, i.e., 1 g of hydrogen ( ![]() Ā) contains 6.023 ū 1023 atoms.
Ā) contains 6.023 ū 1023 atoms.
![]() ∴1000 g ofĀĀĀĀĀĀ contains 6.023
ū 1023 ū 1000 atoms.
∴1000 g ofĀĀĀĀĀĀ contains 6.023
ū 1023 ū 1000 atoms.
![]()
![]() Within the sun, fourĀĀĀĀĀĀ nuclei combine and form oneĀĀĀĀĀĀĀĀ nucleus. In this process 26 MeV of energy is released.
Within the sun, fourĀĀĀĀĀĀ nuclei combine and form oneĀĀĀĀĀĀĀĀ nucleus. In this process 26 MeV of energy is released.
![]() Hence, the energy released
from the fusion of 1 kgĀĀĀĀĀĀĀ is:
Hence, the energy released
from the fusion of 1 kgĀĀĀĀĀĀĀ is:
![]() Amount ofĀĀĀĀĀĀĀĀ = 1 kg = 1000 g
Amount ofĀĀĀĀĀĀĀĀ = 1 kg = 1000 g
![]() 1 mole, i.e., 235 g ofĀĀĀĀĀĀĀĀ contains 6.023
ū 1023 atoms.
1 mole, i.e., 235 g ofĀĀĀĀĀĀĀĀ contains 6.023
ū 1023 atoms.
![]() ∴1000 g ofĀĀĀĀĀĀĀ contains
∴1000 g ofĀĀĀĀĀĀĀ contains ![]()
It is known that the amount of energy released in the fission of one atom of MeV.
![]() Hence, energy released from the fission of 1 kg ofĀĀĀĀĀĀĀĀĀ is:
Hence, energy released from the fission of 1 kg ofĀĀĀĀĀĀĀĀĀ is:
∴ ![]()
![]() is 200
is 200
Therefore, the energy released in the fusion of 1 kg of hydrogen is nearly 8 times the energy released in the fission of 1 kg of uranium.
![]()
Question 13.31:
Suppose India had a target of producing by 2020 AD, 200,000 MW of electric power, ten percent of which was to be obtained from nuclear power plants. Suppose we are given that, on an average, the efficiency of utilization (i.e. conversion to electric energy) of thermal energy produced in a reactor was 25%. How much amount of fissionable uranium would our country need per year by 2020? Take the heat energy per fission of 235U to be about 200MeV.
Answer
Amount of electric power to be generated, P = 2 ū 105 MW
10% of this amount has to be obtained from nuclear power plants.
![]() ∴Amount of nuclear power,
∴Amount of nuclear power,
= 2 ū 104 MW
= 2 ū 104 ū 106 J/s
= 2 ū 1010 ū 60 ū 60 ū 24 ū 365 J/y
Heat energy released per fission of a 235U nucleus, E = 200 MeV Efficiency of a reactor = 25%
Hence, the amount of energy converted into the electrical energy per fission is calculated as:
Number of atoms required for fission per year:
1 mole, i.e., 235 g of U235 contains 6.023 ū 1023 atoms.
∴Mass of 6.023 ū 1023 atoms of U235 = 235 g = 235 ū 10−3 kg
∴Mass of 78840 ū 1024 atoms of U235
Hence, the mass of uranium needed per year is 3.076 ū 104 kg.
![]()